10377. Betti Base
Nomenclature
CAS number: 481-82-3
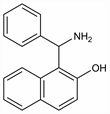
1-(Aminophenylmethyl)-2-naphthalenol; 1-(α-aminobenzyl)-2-naphthol; β-naphthol-phenylaminomethane. C17H15NO; mol wt 249.31.
C 81.90%, H 6.06%, N 5.62%, O 6.42%.
Description and references
The single enantiomers and their derivatives serve as chiral auxiliaries and ligands in asymmetric synthesis. Prepn: M. Betti, Gazz. Chim. Ital. 31, I, 377 (1901). Large scale prepn: idem, Org. Synth. coll. vol. I, 381 (1941). Resolution procedures: idem, Gazz. Chim. Ital. 36, II, 392 (1906); Y. Dong et al., J. Org. Chem. 70, 8617 (2005) DOI PubMed; and absolute configuration determn: C. Cardellicchio et al., Tetrahedron: Asymmetry 9, 3667 (1998) DOI. Synthetic applications: eidem, Tetrahedron 55, 14685 (1999) DOI; Y. Dong et al., Tetrahedron: Asymmetry 15, 1667 (2004) DOI; X. Wang et al., J. Org. Chem. 70, 1897 (2005) DOI PubMed. Review of chemistry of derivatives: I. Szatmári, F. Fülp, Curr. Org. Synth. 1, 155-165 (2004) DOI.
Properties
mp 124-125°.Derivative
Hydrochloride.Nomenclature
CAS number: 219897-32-2
C17H15NO.HCl; mol wt 285.77.
C 71.45%, H 5.64%, N 4.90%, O 5.60%, Cl 12.41%.
Properties
Light pink or white needles, mp 190-220° (dec).Derivative
(R)-Form.Nomenclature
CAS number: 219897-35-5
Properties
mp 137°. [α]D30 60±0.02°.Derivative
(S)-Form.Nomenclature
CAS number: 219897-38-8