12-00731. α-Antiarin [Archived]
Nomenclature
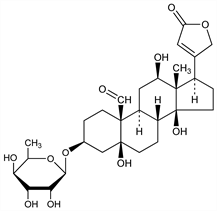
CAS number: 23605-05-2
(3β,5β,12β)-3-[(6-Deoxy-β-d-gulopyranosyl)oxy]-5,12,14-trihydroxy-19-oxocard-20(22)-enolide. C29H42O11; mol wt 566.64.
C 61.47%, H 7.47%, O 31.06%.
Description and references
From latex (arrow poison) of the upas tree, Antiaris toxicaria Lesch., Moraceae, found in Indonesia: Taylor, Br. J. Pharmacol. 8, 237 (1953). Older literature and isoln: Doebel et al., Helv. Chim. Acta 31, 688 (1948); Dolder et al., ibid. 38, 1364 (1955). Structure of aglycone: Martin, Tamm, ibid. 42, 696 (1959). The sugar portion of α-antiarin is antiarose (d-gulomethylose), while that of the isomeric β-antiarin is l-rhamnose. Structure: Juslén et al., ibid. 45, 2285 (1962). Toxicity: Chen, Henderson, J. Pharmacol. Exp. Ther. 150, 53 (1965).
Properties
Tetrahydrate, crystals (six-sided leaflets) from water. Anhydrous after 3 hrs at 110° in high vac, dec 238-240°. [α]D17 3.9° (c = 0.905 in methanol). Sol in water, alcohol, slightly in ether. uv max: 305, 217 nm (log ε 1.8, 4.08). MLD (anhydr compd) in cats (mg/kg): 0.116 i.v. (Chen, Henderson).Derivative
Tribenzoate.C50H54O14; mol wt 878.96.
C 68.32%, H 6.19%, O 25.48%.
Properties
Needles from methanol, mp 240°. [α]D21 +3.0° (acetone). Practically insol in water, very sparingly sol in methanol, ethanol, ether. Freely sol in acetone, chloroform.Status
This monograph has been retired and is no longer subject to revision or update.