12-00912. Aurantiogliocladin [Archived]
Nomenclature
CAS number: 483-54-5
2,3-Dimethoxy-5,6-dimethyl-2,5-cyclohexadiene-1,4-dione; 2,3-dimethoxy-5,6-dimethyl-p-benzoquinone. C10H12O4; mol wt 196.20.
C 61.22%, H 6.16%, O 32.62%.
Description and references
Antibiotic substance produced by a Gliocladium sp similar to Gliocladium roseum (Link.) Bainier: Brian et al., Experientia 7, 266 (1951). Structure: Vischer, J. Chem. Soc. 1953, 815. Synthesis: Baker et al., ibid. 820; Seshadri, Venkatasubramanian, ibid. 1959, 1660. Biosynthesis: Pettersson, Acta Chem. Scand. 19, 1827 (1965).
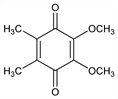
Properties
Bright orange plates from petr ether, mp 62.5°. Absorption max: 407, 275 nm (ε 1200, 38400). Sol in chloroform, ethanol, pyridine. Gives a violet color with concd H2SO4.Derivative
Dihydro deriv.Nomenclature
CAS number: 4373-40-4
(5S-trans)-2,3-Dimethoxy-5,6-dimethylcyclohex-2-ene-1,4-dione; gliorosein. C10H14O4; mol wt 198.22.
C 60.59%, H 7.12%, O 32.29%.
Description and references
Structure: Grave, J. Chem. Soc. C 1966, 985.
Properties
Needles, mp 48°. [α]D22 +125° (c = 0.2 in CHCl3). uv max: 289 nm (ε 24320).Derivative
Quinhydrone deriv.Nomenclature
CAS number: 4516-93-2
Rubrogliocladin. Properties
Dark red needles from petr ether, mp 74°. Absorption max (alcohol): 407, 275 nm (ε 650, 22000). Sol in most organic solvents, water.Status
This monograph has been retired and is no longer subject to revision or update.