12-04384. Gaultherin [Archived]
Nomenclature
CAS number: 490-67-5
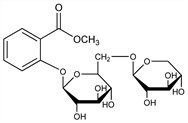
2-[(6-O-β-d-Xylopyranosyl-β-d-glucopyranosyl)oxy]benzoic
acid methyl ester; methyl salicylate-2-glucoxyloside; methyl salicylate-2-primeveroside; monotropitin; monotropitoside. C19H26O12; mol wt 446.40.
C 51.12%, H 5.87%, O 43.01%.
Description and references
In the wintergreen plant, Gaultheria procumbens L., in Monotropa hypopitys L., Ericaceae in Betula lenta L., Betulaceae, in Spiraea ulmaria L. and S. filipendula L., Rosaceae: Bridel, Compt. Rend. 177, 642 (1923); 179, 991 (1924); 180, 1421, 1864 (1925); Bridel, Grillon, ibid. 187, 609 (1928). Synthesis: Robertson, Waters, J. Chem. Soc. 1931, 1881. On hydrolysis with 3% H2SO4 gaultherin forms 1 mol methyl salicylate, 1 mol d-glucose and 1 mol d-xylose. Enzymatic hydrolysis gives methyl salicylate and primeverose (glucoxylose).
Properties
Needles in star formation from 99% acetone. mp 180°. [α]D20 58° (c = 2). Sol in water and alcohol, slightly sol in ethyl acetate, acetone. Insol in ether.Derivative
Gaultherioside.Nomenclature
Ethyl primeveroside.C13H24O10; mol wt 340.32.
C 45.88%, H 7.11%, O 47.01%.
Properties
mp 185°. [α]D 58°. Gaultherioside forms glucose, xylose, and EtOH on hydrolysis with 3% H2SO4.Note
The older literature regards gaultherin as methylsalicylate-d-glucoside.Status
This monograph has been retired and is no longer subject to revision or update.