12-04444. Glaucarubin [Archived]
Nomenclature
CAS number: 1448-23-3
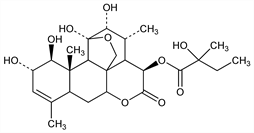
[1β,2α,11β,12α,15β(S)]-11,20-Epoxy-1,2,11,12-tetrahydroxy-15-(2-hydroxy-2-methyl-1-oxobutoxy)picras-3-en-16-one; 2-hydroxy-2-methylbutyric acid 4-ester with 1,2,3,3aβ,4,6aβ,7,7aα,10,11,11a,11bα-dodecahydro-1α,2α,4β,10α,11β-pentahydroxy-3α,8,11aβ-trimethyl-5H-1,11cβ-(epoxymethano)phenanthro[10,1-bc]pyran-5-one; α-kirondrin. C25H36O10; mol wt 496.55.
C 60.47%, H 7.31%, O 32.22%.
Description and references
Antiamebic compound isolated from seeds of Simaruba glauca DC, Simarubaceae: E. A. Ham et al., J. Am. Chem. Soc. 76, 6066 (1954); H. M. Shafer, US 2864745 (1958 to Merck & Co.). Pharmacology: A. C. Cuckler et al., Arch. Int. Pharmacodyn. 114, 307 (1958). Structure: Polonsky et al., Bull. Soc. Chim. Fr. 1964, 1827. Identity of α- and β-kirondrin with glaucarubinone and glaucarubin, resp: Bourguignon, Polonsky, Bull. Soc. Chim. Biol. 46, 1145 (1964). Crystal structure: Kartha et al., Nature 202, 389 (1964). Configuration: Nyburg et al., Chem. Commun. 1965, 203.
Properties
Bitter crystals from methanol, dec 250-255°. [α]D25 +45° (c = 1.7 in pyridine); [α]D25 +69° (c = 0.6 in methanol). Sol in 0.1N NaOH soln. Practically insol in aq sodium bicarbonate solns. Only slightly sol in water (less than 1.8 mg/ml). Prompt acidification of an alkaline soln regenerates glaucarubin, but standing in alkaline soln leads to decompn.Derivative
Tetramethoxyglaucarubin.C29H44O10; mol wt 552.65.
C 63.03%, H 8.02%, O 28.95%.
Properties
Crystals from ether, mp 202-205°. [α]D +17.7° (c = 0.705 in pyridine); [α]D +65.5° (c = 0.926 in methanol).Derivative
Pentaacetate.C35H46O15; mol wt 706.73.
C 59.48%, H 6.56%, O 33.96%.
Properties
Crystals from benzene + petr ether, mp 158-160°. [α]D +49° (c = 0.534 in pyridine).Status
This monograph has been retired and is no longer subject to revision or update.