13-04447. Glafenine [Archived]
Nomenclature
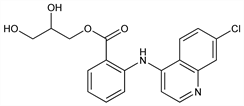
CAS number: 3820-67-5
2-[(7-Chloro-4-quinolinyl)amino]benzoic acid 2,3-dihydroxypropyl
ester; N-(7-chloro-4-quinolyl)anthranilic acid 2,3-dihydroxypropyl
ester; 4-[(2-carboxyphenyl)amino]-7-chloroquinoline α-monoglyceride; 2-[(7-chloro-4-quinolyl)amino]benzoic acid α-glyceride; 2,3-dihydroxypropyl N-(7-chloro-4-quinolyl)anthranilate; glycerylaminophenaquine; glaphenine; R-1707; Privadol (Sanofi-Synthelabo). C19H17ClN2O4; mol wt 372.80.
C 61.21%, H 4.60%, Cl 9.51%, N 7.51%, O 17.17%.
Description and references
Prepn: FR M2413; A. Allais, J. Meier, US 3232944 (1964, 1966 both to Roussel-UCLAF); G. Mouzin et al., Synthesis 1980, 54. HPLC determn in plasma: M.-C. Tournet et al., J. Chromatogr. 224, 348 (1981). Biotransformation in the rat and man: J. Pottier et al., Eur. J. Drug Metab. Pharmacokinet. 4, 109 (1979). Bioavailability, absorption: F. Moolenaar et al., Int. J. Pharm. 4, 195 (1980). Alkalimetric determn: M. S. Tawakkol et al., Pharmazie 36, 163 (1981). Comprehensive description: A. A. Badwan et al., Anal. Profiles Drug Subs. Excip. 21, 197-232 (1992).
Properties
Pale yellow prisms, mp 169-170° (Mouzin); also reported as mp 165° (Allais, Meier). Practically insol in water. Slightly sol in abs alcohol, acetone, ether, benzene, chloroform; sol in dil aq alkalies and acids. Soly at 30°C (g/100 ml): hexane <0.001, water 0.001, chloroform 0.260, acetone 0.297, ethanol 0.700, 0.1N HCl 1.295. pKa (20°C) 7.2. uv max (methanol): 356, 225, 255 nm; (0.1N HCl) 342, 223, 252 nm. LD50 orally in mice: >2 g/kg (Allais, Meier).Therapeutic Category
Analgesic.
Keywords
Analgesic (Non-Narcotic)
Status
This monograph has been retired and is no longer subject to revision or update.