12-04762. Holarrhenine [Archived]
Nomenclature
CAS number: 561-22-8
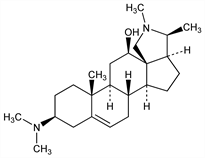
(3β,12β)-3-(Dimethylamino)con-5-enin-12-ol; 12β-hydroxyconessine. C24H40N2O; mol wt 372.59.
C 77.37%, H 10.82%, N 7.52%, O 4.29%.
Description and references
From leaves and bark of Holarrhena congolensis Stapf, Apocynaceae. Isoln: Pyman, J. Chem. Soc. 115, 163 (1919). Structure: Uffer, Helv. Chim. Acta 39, 1834 (1956); van Hove, Tetrahedron 7, 104 (1959).
Properties
Needles from acetic acid or ethyl acetate, mp 197-198°. [α]D24 7° (chloroform); [α]D24 +9° (alcohol). Practically insol in water. Sol in alcohol, chloroform, slightly in ether, acetone, ethyl acetate.Derivative
O-Acetylholarrhenine.C26H42N2O2; mol wt 414.62.
C 75.32%, H 10.21%, N 6.76%, O 7.72%.
Properties
Crystals from pentane or acetone, mp 177-178°.Status
This monograph has been retired and is no longer subject to revision or update.