12-05246. β-Isosparteine [Archived]
Nomenclature
CAS number: 24915-04-6
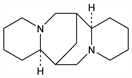
[7S-(7α,7aβ,14α,14aβ)]-Dodecahydro-7,14-methano-2H,6H-dipyrido[1,2-a:1′,2′-e][1,5]diazocine; pusilline. C15H26N2; mol wt 234.38.
C 76.87%, H 11.18%, N 11.95%.
Description and references
Isoln from Lupinus pusillus, Pursh., Leguminosae: Marion, Fenton, J. Org. Chem. 13, 780 (1948). Identity with pusilline: Greenhalgh, Marion, Can. J. Chem. 34, 456 (1956); identity of monohydrochloride with nonalupine and spathulatine: Marion, ibid. 29, 959 (1951). Synthesis: Bohlmann, Ber. 90, 653 (1957). Absolute configuration: Okuda, Tsuda, Chem. Ind. (London) 1961, 1115.
Properties
Oil, bp0.2 100-110°. [α]D32 15.3° (c = 2.3 in abs alc). Sol in methanol, ethanol. Practically insol in water.Derivative
Hydrochloride monohydrate.C15H26N2.HCl.H2O; mol wt 288.86.
C 62.37%, H 10.12%, N 9.70%, Cl 12.27%, O 5.54%.
Properties
Prismatic needles from alcohol or acetone. Melts at 88-90°, resolidifies, and remelts at 235.5-236.5°. [α]D 1.88° (c = 5.90 in chloroform); 2.32° (c = 6.42 in alc); 2.44° (c = 6.42 in water). Sol in water, benzene, alcohol, chloroform, carbon tetrachloride, acetone, ethyl acetate; slightly sol in petr ether, ether. The aq soln is slightly alkaline to litmus.Derivative
Dihydrochloride.C15H26N2.2HCl; mol wt 307.30.
C 58.63%, H 9.18%, N 9.12%, Cl 23.07%.
Properties
Clusters of fine needles from ethyl acetate, mp 270-273° (sinters at 262°).Derivative
Hydriodide.C15H26N2.HI; mol wt 362.29.
C 49.73%, H 7.51%, N 7.73%, I 35.03%.
Properties
Prismatic needles from acetone, mp 252.5° (sinters at 250.5°).Derivative
Methiodide.C15H26N2.CH3I; mol wt 376.32.
C 51.07%, H 7.77%, N 7.44%, I 33.72%.
Properties
Diamond-shaped crystals from ethyl acetate, mp 250-252°.Status
This monograph has been retired and is no longer subject to revision or update.