12-05578. Lobelanine [Archived]
Nomenclature
CAS number: 579-21-5
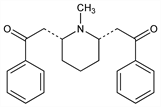
cis-2,2′-(1-Methyl-2,6-piperidinediyl)bis[1-phenylethanone]; 2,2′′-(1-methyl-2,6-piperidinediyl)diacetophenone. C22H25NO2; mol wt 335.44.
C 78.77%, H 7.51%, N 4.18%, O 9.54%.
Description and references
After lobeline, the most abundant alkaloid of Lobelia inflata L., Lobeliaceae. Isoln: Wieland et al., Ann. 444, 40 (1925). Synthesis: Schpf, Lehmann, ibid. 518, 1 (1935); Parker et al., J. Chem. Soc. 1959, 2433.
Properties
Rosettes of needles from petr ether or ether, mp 99°. Freely sol in alcohol, acetone, benzene, chloroform; slightly sol in water, ether.Derivative
Hydrochloride.C22H25NO2.HCl; mol wt 371.90.
C 71.05%, H 7.05%, N 3.77%, O 8.60%, Cl 9.53%.
Properties
Crystals from dil alcohol, dec 188°. Sol in chloroform; slightly sol in abs alcohol, cold water.Derivative
Hydrobromide.C22H25NO2.HBr; mol wt 416.35.
C 63.46%, H 6.29%, N 3.36%, O 7.69%, Br 19.19%.
Properties
Dec 188°.Derivative
Nitrate.C22H25NO2.HNO3; mol wt 398.45.
C 66.32%, H 6.58%, N 7.03%, O 20.08%.
Properties
mp 153-154°.Status
This monograph has been retired and is no longer subject to revision or update.