12-07264. Penicillin F [Archived]
Nomenclature
CAS number: 118-53-6
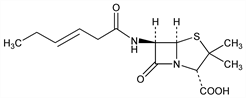
(2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[(1-oxo-3-hexenyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 2-pentenylpenicillin; penicillin I. C14H20N2O4S; mol wt 312.38.
C 53.83%, H 6.45%, N 8.97%, O 20.49%, S 10.26%.
Description and references
Produced by certain strains of Penicillium chrysogenum and P. notatum. Separation and purification by partition chromatography, cf. monograph by Boon and Carrington in Chemistry of Penicillin (Princeton, 1949). Prepn of cryst penicillin F: Wintersteiner, Adler, US 2485227 (1949 to Squibb).
Derivative
Sodium salt.Nomenclature
CAS number: 525-86-0
C14H19N2NaO4S; mol wt 334.37.
C 50.29%, H 5.73%, N 8.38%, Na 6.88%, O 19.14%, S 9.59%.
Properties
Crystals. A trihydrate and a sesquihydrate have been obtained. The anhydr form exists in two cryst modifications. Usually long, fine, blunt-ended needles from water + butanol. After drying in vacuo over P2O5 and then at 55-60° in high vacuum: mp 204-205° (with decompn, open capillary in block preheated to 200°). [α]D25 +316° (c = 0.88). pK in water at 5° = 2.83; at 25° = 2.87. Very sol in water, in isotonic sodium chloride soln and in glucose solns; also sol in alcohol, but is inactivated by this solvent, likewise by glycerol and other primary alcohols. Insol in benzene, carbon tetrachloride, liquid petrolatum.Status
This monograph has been retired and is no longer subject to revision or update.