12-08232. Quinamine [Archived]
Nomenclature
CAS number: 464-85-7
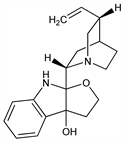
[1S-[1α,2α(3aS*,8aR*),4α,5β]]-8a-(5-Ethenyl-1-azabicyclo[2.2.2]oct-2-yl)-2,3,8,8a-tetrahydro-3aH-furo[2,3-b]indol-3a-ol. C19H24N2O2; mol wt 312.41.
C 73.05%, H 7.74%, N 8.97%, O 10.24%.
Description and references
In Cinchona barks, especially those of Cinchona pubescens Vahl. (C. succirubra Pav.) and C. officinalis L. (C. ledgeriana Moens), Rubiaceae: Hesse, Ber. 5, 265 (1872); 10, 2157 (1877); Ann. 207, 288 (1881); Oudemans, Ann. 197, 49 (1879). Isoln from quinine sulfate mother liquors: De Vrij, Pharm. J. 4, 609 (1874); Howard, ibid. 5, 1 (1875); Henry et al., J. Chem. Soc. 1945, 524. Reduction with lithium aluminum hydride yields cinchonamine: Goutarel et al., Helv. Chim. Acta 33, 150 (1950). Structure: Witkop, J. Am. Chem. Soc. 72, 2311 (1950).
Properties
Needles from boiling benzene, mp 185-186°. [α]D +116° or +104° (c = 0.5 in alcohol). uv max (methanol): 242, 301 nm (log ε 3.93, 3.39). One gram dissolves in 1500 ml water, 50 ml ether, about 120 ml 80% alcohol. Sol in abs alc, hot ether, hot benzene, hot petr ether.Derivative
Hydrochloride monohydrate.C19H25ClN2O2.H2O; mol wt 366.88.
C 62.20%, H 7.42%, Cl 9.66%, N 7.64%, O 13.08%.
Properties
Crystals, mp 166-167°. [α]D +102.8°.Derivative
Hydriodide.C19H25IN2O2; mol wt 440.32.
C 51.83%, H 5.72%, I 28.82%, N 6.36%, O 7.27%.
Properties
Crystals, mp 224°. [α]D +84.82° (c = 0.5 in alcohol).Derivative
Nitrate.C19H25N3O5; mol wt 375.42.
C 60.79%, H 6.71%, N 11.19%, O 21.31%.
Properties
Crystals, mp 186-188°. [α]D +94.9°.Status
This monograph has been retired and is no longer subject to revision or update.