13-02374. Clidanac [Archived]
Nomenclature
Description and references
Prepn: P. F. Juby et al., DE 2004038; eidem, US 3565943 (1970, 1971 both to Bristol-Myers); S. Noguchi et al., Chem. Pharm. Bull. 19, 646 (1971). Synthesis and anti-inflammatory activity: eidem, ibid. 22, 529 (1974); G. R. Allen et al., J. Med. Chem. 15, 934 (1972); and toxicity data: P. F. Juby et al., ibid. 1297. Pharmacology: S. Kuzuna et al., Jpn. J. Antibiot. 24, 695 (1974), C.A. 82, 80297h (1976). X-ray crystallographic study: K. Kamiya et al., Chem. Pharm. Bull. 23, 1589 (1975). Metabolism: S. Tanayama, Y. Kanai, Xenobiotica 7, 145 (1977). Immunogenicity study: S. Kuzuna et al., Oyo Yakuri 19, 951 (1980), C.A. 94, 76682 (1981). Inhibition of prostaglandin biosynthesis: S. Tamura et al., J. Pharm. Pharmacol. 33, 29 (1981). Antipyretic effect in cancer patients: H. Furue, Curr. Ther. Res. 30, 71 (1981).
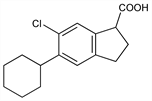
Properties
Colorless crystals from petr ether, mp 150.5-152.5°. LD50 orally in rats: 41 mg/kg (Juby, 1972).Derivative
(S)-(+)-Form.Properties
Colorless needles from petr ether, mp 135-136°. [α]D25 28.7°; [α]25365 +87.7° (c = 2 in ethanol). LD50 orally in rats: 35 mg/kg (Juby, 1972).Derivative
(R)-()-Form.Properties
Colorless cryst from petr ether, mp 134-135°. [α]D25 28.2°; [α]25365 87.5° (c = 2 in ethanol).Therapeutic Category
Anti-inflammatory; antipyretic.
Keywords
Anti-inflammatory (Nonsteroidal); Arylcarboxylic Acids; Antipyretic
Status
This monograph has been retired and is no longer subject to revision or update.