13-03233. Dimepheptanol [Archived]
Nomenclature
CAS number: 545-90-4
β-[2-(Dimethylamino)propyl]-α-ethyl-β-phenylbenzeneethanol; 6-(dimethylamino)-4,4-diphenyl-3-heptanol; 2-dimethylamino-4,4-diphenyl-5-heptanol; 3-hydroxy-4,4-diphenyl-6-dimethylaminoheptane; bimethadol; Methadol; Pangerin. C21H29NO; mol wt 311.46.
C 80.98%, H 9.38%, N 4.50%, O 5.14%.
Description and references
Prepn of α-isomers by catalytic or lithium aluminum hydride reduction of methadone: Speeter et al., J. Am. Chem. Soc. 71, 57 (1949); Pohland et al., ibid. 460. Prepn of β-isomers by sodium propanol reduction of methadone: Eddy et al., J. Org. Chem. 17, 321 (1952); Clark, US 2565592 and US 2668814 (1951 and 1954 to Merck & Co.); Speeter, US 2649445 (1953 to Bristol). Stereochemistry: Portoghese, Williams, 152nd Am. Chem. Soc. Meet. (New York, Sept. 1966) Abstracts of Papers, p 5.
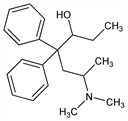
Derivative
β-dl-Form.Properties
mp 127-128°.Derivative
β-dl-Form Hydrochloride.Properties
Rods from acetone, mp 210-212°.Derivative
β-d-Form.Properties
Prisms from alc + water, mp 106-107°; [α]D20 +178° (c = 0.63 in alc).Derivative
β-d-Form Hydrochloride.Properties
Plates from acetone + ether, mp 206-208°. [α]D20 +73.9° (c = 0.69).Derivative
β-l-Form.Properties
Prisms, mp 105-107°. [α]D20 178° (c = 1.04 in alc).Derivative
β-l-Form Hydrochloride.Properties
mp 206-208°. [α]D20 74° (c = 0.94).Derivative
α-dl-Form Hydrochloride.Properties
mp 192-193°.Derivative
α-d-Form Hydrochloride.Properties
mp 169-171°. [α]D25 +34° (c = 0.26).Derivative
α-l-Form Hydrochloride.Properties
mp 169-171°. [α]D25 35° (c = 0.30).Note
This is a controlled substance (opiate): 21 CFR, 1308.11.Therapeutic Category
Analgesic (narcotic).
Keywords
Analgesic (Narcotic)
Status
This monograph has been retired and is no longer subject to revision or update.