Action and use
Cyclo-oxygenase inhibitor; analgesic; anti-inflammatory.
Definition
Fenbufen Tablets contain Fenbufen.
The tablets comply with the requirements stated under Tablets and with the following requirements.
Content of fenbufen, C16H14O3
95.0 to 105.0% of the stated amount.
Identification
A. To a quantity of the powdered tablets containing 0.9 g of Fenbufen add 10 mL of
acetone, triturate using a glass pestle, filter through filter paper wetted with
acetone into 100 mL of
petroleum spirit (boiling point, 40° to 60°), stir rapidly with a glass rod to induce crystallisation and allow to stand for 15 minutes. Filter through a fine porosity sintered-glass funnel, rinse the crystals with 25 mL of
petroleum spirit (boiling point, 40° to 60°), remove the solvent under reduced pressure and dry the crystals at 105° for 15 minutes. The
infrared absorption spectrum of the dried crystals,
Appendix II A, is concordant with the
reference spectrum of fenbufen
(RS 140).
B. In the Assay, the retention time of the principal peak in the chromatogram obtained with solution (1) is the same as that of the peak in the chromatogram obtained with solution (2).
Tests
Dissolution
Comply with the requirements for Monographs of the British Pharmacopoeia in the dissolution test for tablets and capsules, Appendix XII B1.
test conditions
(a) Use Apparatus 2, rotating the paddle at 100 revolutions per minute.
procedure
(1) After 45 minutes withdraw a 10 mL sample of the medium, filter (Whatman 541 paper is suitable) and dilute 1 mL of the filtrate to 50 mL with the dissolution medium. Measure the
absorbance of this solution,
Appendix II B, at 285 nm using dissolution medium in the reference cell.
(2) Measure the
absorbance of a suitable solution of
fenbufen BPCRS prepared by dissolving 50 mg in 50 mL of
methanol and diluting to volume with the dissolution medium and using dissolution medium in the reference cell.
determination of content
Calculate the total content of fenbufen, C16H14O3, in the medium from the absorbances obtained and using the declared content of C16H14O3, in fenbufen BPCRS.
Related substances
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
(1) To a quantity of the powdered tablets containing 0.25 g of Fenbufen add 20 mL of
dimethylformamide, mix with the aid of ultrasound for 20 minutes, add sufficient of the initial mobile phase to produce 50 mL and filter (Whatman GF/F is suitable).
(2) Dilute 1 volume of solution (1) to 100 volumes with the initial mobile phase and further dilute 1 volume of this solution to 10 volumes with the initial mobile phase.
chromatographic conditions
(b) Use gradient elution and the mobile phases described below.
(c) Use a flow rate of 2 mL per minute.
(d) Use an ambient column temperature.
(e) Use a detection wavelength of 254 nm.
(f) Inject 20 µL of each solution.
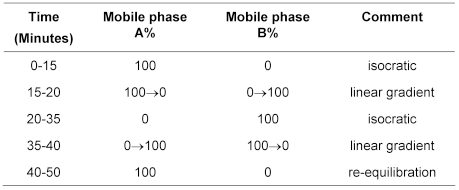
mobile phase
system suitability
The test is not valid unless in the chromatogram obtained with solution (3) the resolution factor between the two principal peaks is at least 10.0.
limits
In the chromatogram obtained with solution (1):
the area of any secondary peak is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.1%);
the sum of the areas of any such peaks is not greater than five times the area of the principal peak in the chromatogram obtained with solution (2) (0.5%).
Assay
Weigh and powder 20 tablets. Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
(1) To a quantity of the powdered tablets containing 0.1 g of Fenbufen add 30 mL of
methanol, mix with the aid of ultrasound for 15 minutes, add sufficient of the mobile phase to produce 100 mL, filter through a 0.45-µm filter (Whatman GF/C is suitable) and dilute 1 volume to 10 volumes with the mobile phase.
(2) Dissolve
fenbufen BPCRS in
methanol, add sufficient of the mobile phase to produce a 0.1% w/v solution and dilute 1 volume of this solution to 10 volumes with the mobile phase.
chromatographic conditions
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 1.5 mL per minute.
(d) Use a column temperature of 40°.
(e) Use a detection wavelength of 280 nm.
(f) Inject 20 µL of each solution.
mobile phase
1 volume of glacial acetic acid, 44 volumes of acetonitrile and 55 volumes of water.
determination of content
Calculate the content of C16H14O3 in the capsules using the declared content of C16H14O3 in fenbufen BPCRS.