Ferrous Fumarate and Folic Acid Tablets
Action and use
Source of iron + Vitamin B component; used in prevention and treatment of anaemias.
Definition
Ferrous Fumarate and Folic Acid Tablets contain Ferrous Fumarate and Folic Acid.
Content of ferrous fumarate, C4H2FeO4
90.0 to 105.0% of the stated amount.
Content of folic acid, C19H19N7O6
90.0 to 115.0% of the stated amount.
Identification
Tests
Dissolution
Comply with the requirements in the dissolution test for tablets and capsules, Appendix XII B1.
For folic acid
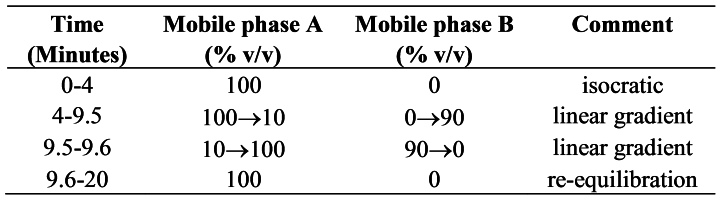
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
Mobile phase A1 volume of formic acid, 100 volumes of methanol and 900 volumes of water.
Mobile phase B1 volume of formic acid, 100 volumes of water and 900 volumes of methanol.
Calculate the total content of C19H19N7O6 in the medium from the chromatograms obtained and using the declared content of C19H19N7O6 in folic acid BPCRS.
The amount of Folic acid released is not less than 75% (Q) of the stated amount.
For ferrous fumarate
After 60 minutes withdraw a sample of the medium and filter. Titrate 100 mL of the filtrate with 0.01M ammonium cerium(IV) sulfate VS using ferroin solution as indicator.
Calculate the total content of C4H2FeO4 in the medium taking each mL of 0.01m ammonium cerium(IV) sulfate VS to be equivalent to 16.99 mg of C4H2FeO4.
The amount of ferrous fumarate released is not less than 75% (Q) of the stated amount.
Ferric iron
Dissolve a quantity of the powder prepared for the Assay for ferrous fumarate containing 1.5 g of Ferrous Fumarate in a mixture of 100 mL of water and 10 mL of hydrochloric acid by heating rapidly to the boiling point. Boil for 15 seconds, cool rapidly, add 3 g of potassium iodide, stopper, allow to stand in the dark for 15 minutes and titrate the liberated iodine with 0.1m sodium thiosulfate VS using starch mucilage as indicator. Repeat the operation without the substance being examined. The difference between the titrations is not more than 13.4 mL (5% ferric iron in Ferrous Fumarate).
Uniformity of Content
For folic acid
Tablets containing less than 2mg and/or less than 2% w/w of Folic Acid comply with the requirements stated under Tablets using the following method of analysis. Carry out the method for liquid chromatography, Appendix III D, using the following solutions in 135 volumes of methanol and 800 volumes of a 0.57% w/v solution of dipotassium hydrogen orthophosphate (solvent A).
135 volumes of methanol and 800 volumes of a solution containing 0.938% w/v of sodium perchlorate and 0.075% w/v of potassium dihydrogen orthophosphate adjusted to pH 7.2 with 0.1m potassium hydroxide and diluted to 1000 volumes with water.
Calculate the content of C19H19N7O6 in each tablet using the declared content of C19H19N7O6 in folic acid BPCRS.
Assay
Weigh and powder 20 tablets.
For ferrous fumarate
Disperse a quantity of the powder containing 0.3 g of Ferrous Fumarate in 7.5 mL of 1m sulfuric acid with gentle heating. Cool, add 25 mL of water and titrate immediately with 0.1m ammonium cerium(iv) sulfate VS using ferroin solution as indicator. Each mL of 0.1m ammonium cerium(iv) sulfate VS is equivalent to 16.99 mg of C4H2FeO4.
For folic acid
For tablets containing less than 2 mg and/or less than 2% w/w of folic acid
Use the average of the individual results obtained in the test for Uniformity of content.
For tablets containing 2 mg or more and 2% w/w or more of folic acid
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in 135 volumes of methanol and 800 volumes of a 0.57% w/v solution of dipotassium hydrogen orthophosphate (solvent A).
135 volumes of methanol and 800 volumes of a solution containing 0.938% w/v of sodium perchlorate and 0.075% w/v of potassium dihydrogen orthophosphate adjusted to pH 7.2 with 0.1m potassium hydroxide and diluted to 1000 volumes with water.
Calculate the content of C19H19N7O6 in the tablets using the declared content of C19H19N7O6 in folic acid BPCRS.
Storage
Ferrous Fumarate and Folic Acid Tablets should be protected from light.
The quantity of the active ingredient is stated both as the amount of ferrous fumarate and in terms of the equivalent amount of ferrous iron.