Morphine Tablets
Action and use
Opioid receptor agonist; analgesic.
Definition
Morphine Tablets contain Morphine Sulfate.
Content of morphine sulfate, (C17H19NO3)2,H2SO4,5H2O
92.5 to 107.5% of the stated amount.
Identification
2.5 volumes of 13.5m ammonia, 32.5 volumes of acetone, 35 volumes of ethanol (70%) and 35 volumes of toluene.
The test is not valid unless the chromatogram obtained with solution (3) shows two clearly separated spots.
The principal spot in the chromatogram obtained with solution (1) corresponds in size, position and colour to that in the chromatogram obtained with solution (2).
Tests
Dissolution
Comply with the requirements for Monographs of the British Pharmacopoeia in the dissolution test for tablets and capsules, Appendix XII B1.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
0.5 volumes of orthophosphoric acid, 300 volumes of acetonitrile and 700 volumes of 0.01m sodium octanesulfonate.
When the chromatograms are recorded under the prescribed conditions, the retention times of morphine sulfate and pseudomorphine are about 2.5 minutes and 3.5 minutes respectively.
The test is not valid unless, in the chromatogram obtained with solution (3), the resolution between the peaks due to morphine and pseudomorphine is at least 4.0.
Calculate the total content of morphine sulfate, (C17H19NO3)2,H2SO4,5H2O in the medium, using the chromatograms obtained and the declared content of (C17H19NO3)2,H2SO4,5H2O in morphine sulfate BPCRS.
Related substances
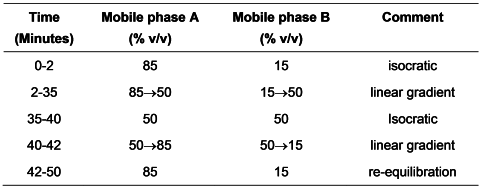
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in solvent A.
Solvent A 1.0% v/v solution of acetic acid.
Mobile phase A0.101% w/v of sodium heptanesulfonate, adjusted to pH 2.6 with a 50% v/v solution of orthophosphoric acid.
Mobile phase Bmethanol.
When the chromatograms are recorded under the prescribed conditions the retention times relative to morphine (retention time about 12.5 minutes) are: impurity F, about 0.95; impurity E, about 1.1; impurity C, impurity A, about 1.4; about 1.6 and impurity B, about 1.9.
The test is not valid unless, in the chromatogram obtained with solution (4), the resolution between the peaks due to impurity F and morphine is at least 4.
Identify any peaks in the chromatogram obtained with solution (1) corresponding to impurity E, impurity C and impurity B using the chromatogram obtained with solution (4) and the chromatogram supplied with morphine for system suitability EPCRS. Multiply the area of any peak corresponding to impurity E, impurity C and impurity B by the following correction factors respectively: 0.5, 0.4 and 0.25.
In the chromatogram obtained with solution (1):
the area of any peak corresponding to impurity B is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (1.0%);
the area of any peak corresponding to impurity A is not greater than half of the area of the principal peak in the chromatogram obtained with solution (2) (0.5%);
the area of any other secondary peaks is not greater than the area of the principal peak in the chromatogram obtained with solution (3) (0.2%);
the sum of the areas of all secondary peaks is not greater than twice the area of the principal peak in the chromatogram obtained with solution (2) (2.0%).
Disregard any peak with an area less than the area of the principal peak in the chromatogram obtained with solution (5) (0.05%).
Assay
Weigh and powder 20 tablets. Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
0.05 volumes of orthophosphoric acid, 30 volumes of acetonitrile and 70 volumes of 0.01m sodium octanesulfonate.
When the chromatograms are recorded under the prescribed conditions, the retention times of morphine sulfate and pseudomorphine are about 2.5 minutes and 3.5 minutes respectively.
The test is not valid unless, in the chromatogram obtained with solution (3), the resolution between the peaks due to morphine and pseudomorphine is at least 4.0.
Calculate the content of (C17H19NO3)2,H2SO4,5H2O in the tablets using the declared content of (C17H19NO3)2,H2SO4,5H2O in morphine sulfate BPCRS.
IMPURITIES
The impurities limited by the requirements of this monograph include those listed under Morphine Sulfate.