Tenoxicam Tablets
Action and use
Cyclo-oxygenase inhibitor; analgesic; anti-inflammatory.
Definition
Tenoxicam Tablets contain Tenoxicam.
Content of tenoxicam, C13H11N3O4S2
92.5 to 105.0% of the stated amount.
Identification
4 volumes of anhydrous formic acid, 30 volumes of acetone and 70 volumes of dichloromethane.
The principal spot in the chromatogram obtained with solution (1) corresponds in position and colour to that in the chromatogram obtained with solution (2).
Tests
Dissolution
Comply with the requirements for Monographs of the British Pharmacopoeia in the dissolution test for tablets and capsules, Appendix XII B1.
Calculate the total content of tenoxicam, C13H11N3O4S2, in the medium from the absorbances obtained and using the declared content of C13H11N3O4S2 in tenoxicam BPCRS.
Related substances
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
Condition the column with the mobile phase for 3 hours.
Dissolve 0.12 g of sodium dodecyl sulfate in 700 mL of methanol, mix with 1000 mL of 0.05m potassium dihydrogen orthophosphate and adjust the pH to 2.8 with orthophosphoric acid.
The test is not valid unless the chromatogram obtained with solution (4) closely resembles the reference chromatogram supplied with tenoxicam degradation impurity standard BPCRS.
the area of any peak corresponding to 2-pyridylamine is not greater than the area of the principal peak in the chromatogram obtained with solution (3) (0.25%).
the area of any secondary peak is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.5%);
the sum of the areas of any such peaks is not greater than four times the principal peak in the chromatogram obtained with solution (2) (2%).
Assay
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
The chromatographic conditions described under Related substances may be used with a detection wavelength of 290 nm.
Dissolve 0.12 g of sodium dodecyl sulfate in 700 mL of methanol, mix with 1000 mL of 0.05m potassium dihydrogen orthophosphate and adjust the pH to 2.8 with orthophosphoric acid.
The test is not valid unless the chromatogram obtained with solution (3) closely resembles the reference chromatogram supplied with tenoxicam degradation impurity standard BPCRS.
Calculate the content of C13H11N3O4S2 in the tablets using the declared content of C13H11N3O4S2 in tenoxicam BPCRS.
IMPURITIES
The impurities limited by the requirements of this monograph include those impurities listed under Tenoxicam and also the following.
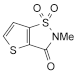
1. 2-methylthieno[2,3-d]isothiazol-3(2 H)-one-1,1-dioxide,
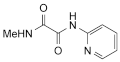
2. N-methyl-N′-(2-pyridyl)oxamide,
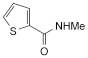
3. N-methylthiophene-2-carboxamide.