459. Aminopentamide
Nomenclature
CAS number: 60-46-8
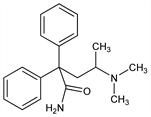
α-[2-(Dimethylamino)propyl]-α-phenylbenzeneacetamide; 4-dimethylamino-2,2-diphenylvaleramide; α,α-diphenyl-γ-dimethylaminovaleramide; dimevamide; BL-139. C19H24N2O; mol wt 296.41.
C 76.99%, H 8.16%, N 9.45%, O 5.40%.
Description and references
Anticholinergic. Prepn similar to that of methadone, starts by condensing diphenylmethyl cyanide with chlorodimethylaminopropane: Walton, et al., J. Chem. Soc. 1949, 648; Cheney et al., J. Org. Chem. 17, 770 (1952); Specter, US 2647926 (1953 to Bristol). Prepn of optical isomers: Wheatley et al., J. Org. Chem. 19, 794 (1954). See also Moffett, Aspergren, J. Am. Chem. Soc. 79, 4451 (1957). Toxicity study: Cazort, J. Pharmacol. Exp. Ther. 100, 325 (1950).
Derivative
dl-Form.Properties
Long prisms from dilute alcohol, mp 183-184°. Practically insol in water.Derivative
d-Form.Properties
Crystals from petr ether, mp 136.5-137.5° (sintering). [α]D23 +98.9° (methanol).Derivative
l-Form.Properties
Crystals from petr ether, mp 136.5-137.5° (sintering). [α]D23 101.9° (methanol).Derivative
dl-Form hydrochloride.C19H24N2O.HCl; mol wt 332.87.
C 68.56%, H 7.57%, N 8.42%, O 4.81%, Cl 10.65%.
Properties
Slightly deliquescent leaflets from alcohol + ether, dec 190-191°. Bitter taste. Soluble in water, alcohol. A 1% aq soln has a pH of 6.8. LD50 in mice (mg/kg): 34.7 i.v.; 396 orally (Cazort).Derivative
dl-Form acid sulfate.Nomenclature
Centrine (Fort Dodge).C19H24N2O.H2SO4; mol wt 394.49.
C 57.85%, H 6.64%, N 7.10%, O 20.28%, S 8.13%.
Properties
Deliquescent crystals from isopropanol + ethyl acetate, mp 185-187°. The commercial medicinal grade, mp 178-181°. Bitter taste. uv max (1% H2SO4): 258.5 nm (A1%1cm 10.3); min: 249 nm. Freely sol in water, alc. Very slightly sol in chloroform. Practically insol in ether. pH of 2.5% aq soln 1.3-2.2.Therapeutic Category
Antispasmodic.
Therapeutic Category (Veterinary)
Antispasmodic; antiemetic.Keywords
Antimuscarinic; Antispasmodic