618. β-Amyrin
Nomenclature
CAS number: 559-70-6
(3β)-Olean-12-en-3-ol; β-amyrenol. C30H50O; mol wt 426.72.
C 84.44%, H 11.81%, O 3.75%.
Description and references
Occurs together with α-amyrin. Isoln and structural studies: see α-amyrin. See also Vesterberg, Bull. Soc. Chim. 37, 742 (1925); Horrmann, Firzlaff, Arch. Pharm. 268, 64 (1930); Ruzicka, Marxer, Helv. Chim. Acta 22, 195 (1939); Jeger, Ruzicka, ibid. 28, 209 (1945); Prelog et al., ibid. 29, 360 (1946). Conversion of δ-amyrene to β-amyrin: Barton et al., J. Chem. Soc. C 1968, 1031. Biogenetic-type total synthesis: van Tamelen et al., J. Am. Chem. Soc. 94, 8229 (1972). Biosynthesis from squalene: Suga et al., Chem. Lett. 1972, 129, 313.
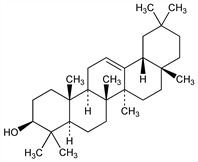
Properties
Needles from petr ether or alc, mp 197-197.5°. bp0.8 260°. [α]D19 +99.8° (c = 1.3 in benzene). Somewhat less soluble than the α-form. Soluble in 37 parts of 98% alc.Derivative
Acetate.C32H52O2; mol wt 468.75.
C 81.99%, H 11.18%, O 6.83%.
Properties
Prisms from petr ether, mp 241°. [α]D17 +79° (c = 0.9 in benzene).Derivative
Palmitate.Nomenclature
Balanophorin.C46H80O2; mol wt 665.13.
C 83.07%, H 12.12%, O 4.81%.
Properties
mp 77°. [α]D15 +54.5° (c = 1.1 in benzene). Occurs in Balanophora elongata Blume, Balanophoraceae, in Erythroxylum coca Lam. var novogranatense Morris, and var spruceanum Burck, Erythroxylaceae, in latex from Ficus variegata Blume, Moraceae.Derivative
Di-β-amyrin ether.C60H98O; mol wt 835.42.
C 86.26%, H 11.82%, O 1.92%.