686. Anthranol
Nomenclature
CAS number: 529-86-2
9-Anthracenol; 9-anthrol; 9-hydroxyanthracene. C14H10O; mol wt 194.23.
C 86.57%, H 5.19%, O 8.24%.
Description and references
Prepd by dissolving anthrone in 5 to 10% boiling NaOH soln, cooling to 5° and pouring in cooled 5% H2SO4: Meyer, Ann. 379, 56 (1911). Also prepared from anthraquinone.
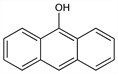
Properties
Needles from glacial acetic acid, plates from dil alcohol. Orthorhombic, mp 120° (if bath is heated to 110°). Sinters at 120°, mp 152° (if bath is cold at start). Absorption spectrum (of acetate): Barnett et al., J. Chem. Soc. 1928, 885. Sol in most solvents with blue fluorescence. The solid is fairly stable when kept dry. In solution it changes quickly to anthrone. Recrystallizations of anthranol may lead to a less pure compound.Derivative
Acetate.C16H12O2; mol wt 236.27.
C 81.34%, H 5.12%, O 13.54%.
Properties
Crystals from petr ether, mp 134°.Derivative
Benzoate.C21H14O2; mol wt 298.33.
C 84.55%, H 4.73%, O 10.73%.
Properties
Crystals from pyridine + alcohol, mp 170-172°.Derivative
Methyl ether.C15H12O; mol wt 208.26.
C 86.51%, H 5.81%, O 7.68%.
Properties
Crystals from alcohol, mp 97-98°.Derivative
Ethyl ether.C16H14O; mol wt 222.28.
C 86.45%, H 6.35%, O 7.20%.