753. Aprepitant
Nomenclature
Description and references
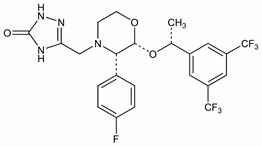
Selective substance P/neurokinin-1 (NK-1) receptor antagonist. Prepn: C. P. Dorn et al., WO 9516679; eidem, US 5719147 (1995, 1998 both to Merck & Co.); and pharmacology: J. J. Hale et al., J. Med. Chem. 41, 4607 (1998). Improved synthesis: K. M. J. Brands et al., J. Am. Chem. Soc. 125, 2129 (2003). LC/MS/MS determn in plasma: C. M. Chavez-Eng et al., J. Pharm. Biomed. Anal. 35, 1213 (2004). Clinical trial in prevention of cisplatin-induced emesis: S. Van Belle et al., Cancer 94, 3032 (2002); with granisetron and/or dexamethasone: D. Campos et al., J. Clin. Oncol. 19, 1759 (2001). Clinical pharmacokinetics: A. K. Majumdar et al., J. Clin. Pharmacol. 46, 291 (2006). Review of clinical experience in prevention of chemotherapy-induced nausea and vomiting (CINV): T. M. Dando, C. M. Perry, Drugs 64, 777-794 (2004); A. M. Massaro, K. L. Lenz, Ann. Pharmacother. 39, 77-85 (2005).
Properties
White to off-white crystalline solid, mp 255°. [α]D25 +69° (c = 1.00 in methanol). Sparingly sol in ethanol, isopropyl acetate; slightly sol in acetonitrile. Practically insol in water.Derivative
Fosaprepitant dimeglumine.Nomenclature
Description and references
Water-soluble prodrug. Prepn: C. P. Dorn et al., WO 9523798; eidem, US 5691336 (1995, 1997 both to Merck & Co.); J. J. Hale et al., J. Med. Chem. 43, 1234 (2000). Bioequivalence to aprepitant and tolerability: K. C. Lasseter et al., J. Clin. Pharmacol. 47, 834 (2007).
Therapeutic Category
Antiemetic.
Keywords
Antiemetic; Neurokinin Receptor Antagonist