770. Arbekacin
Nomenclature
Description and references
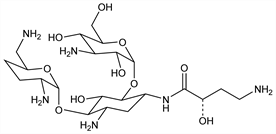
Aminoglycoside antibiotic; derivative of kanamycin B, q.v. Prepn and antibacterial activity: S. Kondo et al., J. Antibiot. 26, 412 (1973); H. Umezawa et al., DE 2350169; eidem, US 4107424 (1974, 1978 both to Microbiochem. Res. Found.). Mechanism of action: N. Tanaka et al., Antimicrob. Agents Chemother. 24, 797 (1983); K. Matsunaga et al., J. Antibiot. 37, 596 (1984). Pharmacology: J. Stewens et al., Arzneim.-Forsch. 35, 1440 (1985); M. Kurebe et al., ibid. 36, 1511 (1986). Series of articles on antibacterial activity, pharmacology, pharmacokinetics, and clinical experience: Chemotherapy 34, Suppl. 1, pp 1-670 (1986). Review: Y. Kobayashi et al., Int. J. Antimicrob. Agents 5, 227-230 (1995).
Derivative
Sulfate.Nomenclature
Derivative
Dicarbonate.Properties
Colorless, crystalline powder, mp 178° (dec). [α]D24 +86.8° (c = 0.77 in water). LD50 i.v. in mice: >150 mg/kg (Umezawa, 1978).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Aminoglycosides