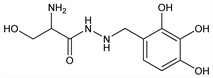
1050. Benserazide
Nomenclature
Description and references
Peripheral decarboxylase inhibitor. Prepn: BE 619015; Hegedüs, Zeller, US 3178476 (1962, 1965 both to Hoffmann-La Roche). Inhibition of dopa decarboxylase in vitro and in vivo: W. P. Burkard et al., Experientia 18, 411 (1962); eidem, Arch. Biochem. Biophys. 107, 187 (1964). Synergy with dopa in vivo: G. Bartholini et al., Nature 215, 852 (1967); G. Bartholini, A. Pletscher, J. Pharmacol. Exp. Ther. 161, 14 (1968). Clinical comparison with carbidopa, q.v.: I. Kuruma et al., J. Pharm. Pharmacol. 24, 289 (1972). Clinical and pharmacokinetic profile: M.-H. Marion et al., Adv. Neurol. 45, 493 (1987). Review of pharmacology and clinical efficacy of decarboxylase inhibitors: R. M. Pinder et al., Drugs 11, 329-377 (1976).
Derivative
Hydrochloride.Nomenclature
Properties
White crystalline powder, mp 146-148°. Sol in water.Derivative
Combination with levodopa.Nomenclature
Therapeutic Category
In combination with levodopa as antiparkinsonian.
Keywords
Antiparkinsonian