1064. Benzathine
Nomenclature
CAS number: 140-28-3
N,N′-Bis(phenylmethyl)-1,2-ethanediamine; N,N′-dibenzylethylenediamine; 1,2-bis(benzylamino)ethane; DBED. C16H20N2; mol wt 240.34.
C 79.96%, H 8.39%, N 11.66%.
Description and references
Prepd from N,N′-dibenzenesulfonyl-N,N′-dibenzylethylenediamine by heating with concd HCl in bomb tube at 170-180°: Bleier, Ber. 32, 1829 (1899); from ethylene chloride and benzylamine: Frost et al., J. Am. Chem. Soc. 71, 3842 (1949); by reduction of N,N′-dibenzylideneethylenediamine: DE 98031 (1898 to Schering); Chem. Zentralbl. 1898, II, 743; Van Alphen, Rec. Trav. Chim. 54, 93 (1935); Lob, ibid. 55, 859 (1936); Szabo et al., Antibiot. Chemother. 1, 499 (1951); Rebenstorf, US 2773098 (1956 to Upjohn).
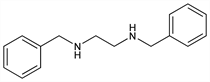
Properties
Oily liquid, mp 26°. d420 1.024. bp12 212-213°; bp4 195°. nD20 1.5624. Insol in water. Freely sol in the usual organic solvents except CS2, with which it gives a solid addition product.Derivative
Diacetate.C20H28N2O4; mol wt 360.45.
C 66.64%, H 7.83%, N 7.77%, O 17.75%.
Properties
Needles, mp 111°. Soly in water: 253 mg/ml.Derivative
Dihydrobromide.C16H22Br2N2; mol wt 402.17.
C 47.78%, H 5.51%, Br 39.74%, N 6.97%.
Properties
Shiny leaflets, dec 300°. Soly in water: 30 mg/ml.Derivative
Dihydrochloride.C16H22Cl2N2; mol wt 313.27.
C 61.34%, H 7.08%, Cl 22.63%, N 8.94%.
Properties
Shiny leaflets, dec 298°. Soly in water: 23.9 mg/ml.Derivative
Sulfate.C16H22N2O4S; mol wt 338.42.
C 56.78%, H 6.55%, N 8.28%, O 18.91%, S 9.47%.
Properties
Crystals, dec 247-250°. Soly in water: 15.8 mg/ml.Derivative
Disalicylate.C30H34N2O6; mol wt 518.60.
C 69.48%, H 6.61%, N 5.40%, O 18.51%.