Nomenclature
CAS number: 65-85-0
Benzenecarboxylic acid; phenylformic acid; dracylic acid.
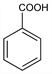
C
7H
6O
2; mol wt 122.12.
C 68.85%, H 4.95%, O 26.20%.
Description and references
Occurs in nature in free and combined forms.
Gum benzoin may contain as much as 20%. Most berries contain appreciable
amounts (around 0.05%). Excreted mainly as hippuric acid by almost
all vertebrates, except fowl. Mfg processes include the air oxidation
of toluene, the hydrolysis of benzotrichloride, and the decarboxylation
of phthalic anhydride: Faith, Keyes &
Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran,
Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 138-144. Lab
prepn from benzyl chloride: A. I. Vogel, Practical Organic Chemistry (Longmans, London, 3rd ed,
1959) p 755; from benzaldehyde: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter,
Berlin, 40th ed, 1961) p 193. Prepn of ultra-pure benzoic acid for
use as titrimetric and calorimetric standard: Schwab, Wicher, J. Res. Natl. Bur. Stand. 25,
747 (1940). Review: A. E. Williams in Kirk-Othmer Encyclopedia of Chemical Technology vol. 3 (Wiley-Interscience, New York, 3rd ed., 1978)
pp 778-792.
Properties
Monoclinic tablets, plates, leaflets. d 1.321 (also reported as 1.266). mp 122.4°. Begins to sublime at ≈100°. bp
760 249.2°; bp
400 227°; bp
200 205.8°; bp
100 186.2°; bp
60 172.8°; bp
40 162.6°; bp
20 146.7°; bp
10 132.1°. Volatile with steam. Flash pt 121°C. pK (25°) 4.19. pH of satd soln at 25°: 2.8. Soly
in water (g/l) at 0° = 1.7; at 10° = 2.1; at 20° = 2.9; at 25° = 3.4;
at 30° = 4.2; at 40° = 6.0; at 50° = 9.5; at 60° = 12.0; at 70° =
17.7; at 80° = 27.5; at 90° = 45.5; at 95° = 68.0. Mixtures of excess
benzoic acid and water form two liquid phases beginning at 89.7°.
The two liquid phases unite at the critical soln temp of 117.2°.
Composition of critical mixture: 32.34% benzoic acid, 67.66% water:
see Ward, Cooper,
J. Phys. Chem. 34, 1484 (1930). One gram dissolves in 2.3 ml cold alc,
1.5 ml boiling alc, 4.5 ml chloroform, 3 ml ether, 3 ml acetone, 30
ml carbon tetrachloride, 10 ml benzene, 30 ml carbon disulfide, 23
ml oil of turpentine; also sol in volatile and fixed oils, slightly
in petr ether. The soly in water is increased by alkaline substances,
such as borax or trisodium phosphate,
see also Sodium Benzoate.
Derivative
Barium salt dihydrate.
Nomenclature
CAS number: 5908-68-9
Barium benzoate.
C
14H
10BaO
4.2H
2O; mol wt 415.58.
C 40.46%, H 3.40%, Ba 33.04%, O 23.10%.
Properties
Nacreous leaflets. Poisonous. Soluble in about 20 parts water; slightly sol in alc.Derivative
Calcium salt trihydrate.
Nomenclature
CAS number: 5743-30-6
Calcium benzoate.
C
14H
10CaO
4.3H
2O; mol wt 336.35.
C 49.99%, H 4.79%, Ca 11.92%, O 33.30%.
Properties
Orthorhombic crystals or powder. d 1.44. Soluble in 25 parts water;
very sol in boiling water.Derivative
Cerium salt trihydrate.
Nomenclature
CAS number: 105546-51-8
Cerous benzoate.
C
21H
15CeO
6.3H
2O; mol wt 557.50.
C 45.24%, H 3.80%, Ce 25.13%, O 25.83%.
Properties
White to reddish-white powder. Sol in hot water
or hot alc.Derivative
Copper salt dihydrate.
Nomenclature
CAS number: 6046-97-5
Cupric benzoate.
C
14H
10CuO
4.2H
2O; mol wt 341.80.
C 49.20%, H 4.13%, Cu 18.59%, O 28.09%.
Properties
Light blue, cryst powder. Slightly soluble in
cold water, more in hot water; sol in alc or in dil acids with separation
of benzoic acid.Derivative
Lead salt monohydrate.
Nomenclature
CAS number: 6080-57-5
Lead benzoate.
C
14H
10O
2Pb.H
2O; mol wt 435.44.
C 38.62%, H 2.78%, O 11.02%, Pb 47.58%.
Properties
Cryst powder. Poisonous. Slightly sol in water.Derivative
Manganese salt tetrahydrate.
Nomenclature
CAS number: 6146-97-0
Manganese benzoate.
C
14H
10MnO
4.4H
2O; mol wt 369.23.
C 45.54%, H 4.91%, Mn 14.88%, O 34.67%.
Properties
Pale-red powder. Sol in water, alc. Also occurs
with 3H2O.Derivative
Nickel salt trihydrate.
Nomenclature
CAS number: 6018-91-3
Nickel benzoate.
C
14H
10NiO
4.3H
2O; mol wt 354.97.
C 47.37%, H 4.54%, Ni 16.53%, O 31.55%.
Properties
Light-green odorless powder. Slightly sol in
water; sol in ammonia; dec by acids.Derivative
Potassium salt trihydrate.
Nomenclature
CAS number: 6100-02-3
Potassium benzoate.
C
7H
5KO
2.3H
2O; mol wt 214.26.
C 39.24%, H 5.17%, K 18.25%, O 37.34%.
Properties
Crystalline powder. Sol in water, alc.Derivative
Silver salt.
Nomenclature
CAS number: 532-31-0
Silver benzoate.
C
7H
5AgO
2; mol wt 228.98.
C 36.72%, H 2.20%, Ag 47.11%, O 13.97%.
Properties
Light-sensitive powder. Sol in 385 parts cold
water, more sol in hot water; very slightly sol in alc.Derivative
Uranium salt.
Nomenclature
CAS number: 532-60-5
Uranyl benzoate.
C
14H
10O
6U; mol wt 512.25.
C 32.83%, H 1.97%, O 18.74%, U 46.47%.
Properties
Yellow powder. Slightly sol in water, alc.Caution
Potential symptoms of overexposure
may include irritation to the skin, eyes, nose, throat, and mucous
membranes.Use
Preserving foods, fats, fruit juices, alkaloidal
solns, etc; manuf benzoates and benzoyl compds, dyes; as a mordant
in calico printing; for curing tobacco. As standard in volumetric
and calorimetric analysis. Pharmaceutic aid (antifungal).
Therapeutic Category (Veterinary)
Has been used with salicylic acid as
a topical antifungal.
