1523. Butethamine
Nomenclature
CAS number: 2090-89-3
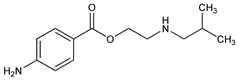
2-[(2-Methylpropyl)amino]ethanol 4-aminobenzoate (ester); 2-(isobutylamino)ethanol p-aminobenzoate (ester); 2-(isobutylamino)ethyl p-aminobenzoate. C13H20N2O2; mol wt 236.31.
C 66.07%, H 8.53%, N 11.85%, O 13.54%.
Description and references
Prepn of the hydrochloride: Goldberg, US 2139818 (1938 to Novocol Chem.); J. Büchi et al., Arzneim.-Forsch. 14, 161 (1964); 16, 1657 (1966).
Derivative
Formate.C13H20N2O2.CH2O2; mol wt 282.34.
C 59.56%, H 7.85%, N 9.92%, O 22.67%.
Properties
mp 136-139°. Freely sol in water and alcohol. Slightly sol in chloroform, ether; very slightly sol in benzene. pH (1% aq soln): about 6.1.Derivative
Hydrochloride.Nomenclature
CAS number: 553-68-4
Ibylcaine; Monocaine (Novocol). C13H20N2O2.HCl; mol wt 272.77.
C 57.24%, H 7.76%, N 10.27%, O 11.73%, Cl 13.00%.
Properties
mp 192-196°. Sol in water, slightly sol in alcohol, chloroform, benzene. Practically insol in ether. pH (1% aq soln): about 4.7.Derivative
meta-Isomer hydrochloride.Nomenclature
CAS number: 553-58-2
Metabutethamine hydrochloride. C13H20N2O2.HCl; mol wt 272.77.
C 57.24%, H 7.76%, N 10.27%, O 11.73%, Cl 13.00%.
Properties
Bitter crystals, mp 181-184°. Sol in water. pH of 1:50 aq soln about 6.2. Slightly sol in alcohol, acetone, chloroform.Therapeutic Category
Anesthetic (local).
Therapeutic Category (Veterinary)
Local anesthetic for nerve block.Keywords
Anesthetic (Local)