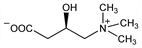
1849. Carnitine
Nomenclature
Description and references
Essential cofactor of fatty acid metabolism; required for the transport of fatty acids through the inner mitochondrial membrane. Synthesized primarily in the liver and kidney; highest concentrations found in heart and skeletal muscle. Name derived from the Latin word, carnis, meaning meat or flesh. Dietary sources include red meat, dairy products, beans, avocado. Isoln from meat extract: W. Gulewitsch, R. Krimberg, Z. Physiol. Chem. 45, 326 (1905); H. E. Carter, P. K. Bhattacharyya, Methods Enzymol. III, 660 (1957). Identification as vitamin BT: H. E. Carter et al., Arch. Biochem. Biophys. 38, 405 (1952). Synthesis: M. Tomita, Y. Sendju, Z. Physiol. Chem. 169, 263 (1927); R. Voeffray et al., Helv. Chim. Acta 70, 2058 (1987). Enantioselective synthesis from glycerol: M. Marzi et al., J. Org. Chem. 65, 6766 (2000). Biosynthesis: G. Wolf, C. R. A. Berger, Arch. Biochem. Biophys. 92, 360 (1961). Absolute configuration: T. Kaneko, R. Yoshida, Bull. Chem. Soc. Jpn. 35, 1153 (1962). Metabolism in humans: M. E. Mitchell, Am. J. Clin. Nutr. 31, 293 (1978). Chromatographic determn in food supplement formulations: A. Kakou et al., J. Chromatogr. A 1069, 209 (2005). Use in canine dilated cardiomyopathy: S. L. Sanderson, Vet. Clin. North Am. Small Anim. Pract. 36, 1325 (2006). Historical review: G. Fraenkel, S. Friedman, Vitam. Horm. XV, 73-118 (1957). Review of physiological significance and deficiency syndromes: C. J. Rebouche, D. J. Paulson, Annu. Rev. Nutr. 6, 41-66 (1986); of clinical pharmacology: J. J. Bahl, R. Bressler, Annu. Rev. Pharmacol. Toxicol. 27, 257-277 (1987); of biosynthesis in mammals: F. M. Vaz, R. J. A. Wanders, Biochem. J. 361, 417-429 (2002). Review of effect on myocardial metabolism and clinical experience in ischemic heart disease: R. Lango et al., Cardiovasc. Res. 51, 21-29 (2001); of use as dietary supplement in athletes: H. Karlic, A. Lohninger, Nutrition 20, 709-715 (2004). Symposium on physiology, pharmacology and therapeutic potential: Ann. N.Y. Acad. Sci. 1033, 1-197 (2004).
Properties
Crystals from anhydrous ethanol + acetone, dec 196-198° (Carter, Bhattacharyya); also reported as crystals from isopropanol, mp 200° (dec) (Marzi). Very hygroscopic solid. [α]D25 -31.3° (c = 10 in water); [α]54625 -37.0° (c = 10 in water). Sol in water, hot alcohol. Practically insol in acetone, ether, benzene.Derivative
Hydrochloride.Nomenclature
Properties
Crystals, dec 142°.Derivative
dl-Form.Nomenclature
Description and references
Synthesis: E. Strack et al., Ber. 86, 525 (1953); H. E. Carter, P. K. Bhattacharyya, J. Am. Chem. Soc. 75, 2503 (1953).
Properties
Hygroscopic crystalline solid, dec 195-197°. Sol in water, ethanol.Derivative
dl-Form hydrochloride.Nomenclature
Properties
Needles from ethanol, mp 196° (dec). Very sol in water; sol in hot ethanol; slightly sol in cold ethanol. Practically insol in acetone, ether.Derivative
d-Form.Nomenclature
Description and references
Prepn: S. Friedman et al., Arch. Biochem. Biophys. 66, 10 (1957).
Properties
Crystals, dec 210-212°. [α]D +30.9°. Very sol in water and alcohol. Practically insol in acetone and ether.Derivative
d-Form hydrochloride.Nomenclature
Properties
Crystals, dec 142°.Therapeutic Category
Vitamin (enzyme cofactor).
Therapeutic Category (Veterinary)
In treatment of feline hepatic lipidosis and canine dilated cardiomyopathy.Keywords
Enzyme Cofactor; Vitamin/Vitamin Source