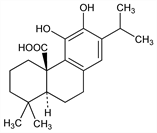
10299. Carnosic Acid
Nomenclature
Description and references
Phenolic diterpene antioxidant principally found in rosemary and sage. Readily cyclizes to form the lactone, carnosol, q.v.; precursor for structurally similar antioxidants found in these plants. Isoln from Salvia officinalis L., Lamiaceae: H. Linde, Helv. Chim. Acta 47, 1234 (1964) DOI; from Rosmarinus officinalis L., Lamiaceae: E. Wenkert et al., J. Org. Chem. 30, 2931 (1965) DOI. Configuration: C. R. Narayanan, H. Linde, Tetrahedron Lett. 6, 3647 (1965) DOI. Antioxidant activity in lipid systems: C. H. Brieskorn, H.-J. Dmling, Z. Lebensm.-Unters. Forsch. 141, 10 (1969) DOI; A. I. Hopia et al. J. Agric. Food Chem. 44, 2030 (1996) DOI. Mechanism of antioxidant action: T. Masuda et al., ibid. 49, 5560 (2001) DOI PubMed. HPLC determn in rosemary extracts and comparison with derivative antioxidant compds: S. L. Richheimer et al., J. Am. Oil Chem. Soc. 73, 507 (1996) DOI. HPLC determn in foods: W. Ternes, K. Schwarz, Z. Lebensm.-Unters. Forsch. 201, 548 (1995) DOI; in antioxidant food packaging material: K. Bentayeb et al., Anal. Bioanal. Chem. 389, 1989 (2007) DOI PubMed.