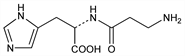
1850. Carnosine
Nomenclature
Description and references
Naturally occurring dipeptide found in large amounts in skeletal muscle. Also present in other tissues such as brain, cardiac muscle, kidney. Water soluble antioxidant; functions as a free-radical scavenger. Isoln: Gulewitsch, Amiradzibi, Ber. 33, 1902 (1900); Wolff, Wilson, J. Biol. Chem. 95, 495 (1932); 109, 565 (1935). Synthesis from histidine and β-iodo- or β-nitropropionyl chloride: Baumann, Ingvaldsen, ibid. 35, 271 (1918); Barger, Tutin, Biochem. J. 12, 406 (1918). Later syntheses: Sifford, du Vigneaud, J. Biol. Chem. 108, 753 (1935); R. A. Turner, J. Am. Chem. Soc. 75, 2388 (1953); F. J. Vinick, S. Jung, J. Org. Chem. 48, 392 (1983). Crystal structure: H. Itoh et al., Acta Crystallogr. 33B, 2959 (1977). Possible role in wound healing: D. E. Fischer et al., Proc. Soc. Exp. Biol. Med. 158, 402 (1978). Review of physiological properties and therapeutic potential: S. E. Gariballa, A. J. Sinclair, Age Ageing 29, 207-210 (2000).