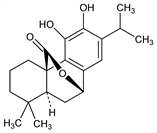
10298. Carnosol
Nomenclature
Description and references
Phenolic diterpene antioxidant principally occurring in rosemary and sage; lactone of carnosic acid, q.v. Isoln from Salvia carnosa, Dougl., Lamiaceae: A. I. White, G. L. Jenkins, J. Am. Pharm. Assoc. 31, 33, 37 (1942). Isoln as picrosalvin from S. officinalis, L.: C. H. Brieskorn, A. Fuchs, Ber. 95, 3034 (1962) DOI. Isoln from rosemary and identity with picrosalvin: idem et al., J. Org. Chem. 29, 2293 (1964) DOI. Prepn by oxidation of carnosic acid: J. G. Marrero et al., J. Nat. Prod. 65, 986 (2002) DOI PubMed. Crystal structure and spectroscopic properties: M. Gajhede et al., J. Crystallogr. Spectrosc. Res. 20, 165 (1990) DOI. Antioxidant activity in lipids: C. H. Brieskorn, H.-J. Dmling, Z. Lebensm.-Unters. Forsch. 141, 1431 (1969) DOI; in biological systems: O. I. Aruoma et al., Xenobiotica 22, 257 (1992) PubMed; in oil-in-water emulsion: E. N. Frankel et al., J. Agric. Food Chem. 44, 131 (1996) DOI. Anticancer effects in murine model of colonic tumorigenesis: A. E. Moran et al., Cancer Res. 65, 1097 (2005) PubMed.