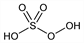
1851. Caro's Acid
Nomenclature
CAS number: 7722-86-3
Peroxymonosulfuric acid; sulfomonoperacid; persulfuric acid. H2O5S; mol wt 114.08.
H 1.77%, O 70.12%, S 28.11%.
Description and references
Dry reagent is prepd by stirring 10 g potassium persulfate into 11 g concd H2SO4 for 10 min and adding 30 g finely powdered potassium sulfate; liquid reagent is obtained by triturating potassium persulfate with three times as much (by weight) of H2SO4; dil reagent is prepd by stirring 10 g potassium persulfate into 11 g concd H2SO4 and adding 50 cc ice: Baeyer, Villiger, Ber. 32, 3625 (1899); another prepn by reacting 90% H2O2 with chlorosulfonic acid at -40 to -50°: Ball, Edwards, J. Am. Chem. Soc. 78, 1125 (1956).