1852. α-Carotene
Nomenclature
Description and references
About as widely distributed as its β-isomer, but in smaller amounts. Best sources for both the α- and β-isomers are carrots, palm oil, and green leaves of various species. As a provitamin A it is half as active as β-carotene. Found in the mother liquors after crystallizing β-carotene. Isoln by chromatography: Karrer, Walker, Helv. Chim. Acta 16, 641 (1933). Structure: Kuhn, Lederer, Ber. 64, 1349 (1931); Karrer et al., Helv. Chim. Acta 14, 614 (1931); 16, 975 (1933). Natural (+)-α-carotene has 6′R configuration: Eugster et al., ibid. 52, 1729 (1969). Synthesis of dl-α-carotene: Eugster, Karrer, ibid. 38, 610 (1955); Tscharner et al., ibid. 40, 1676 (1957); Ruegg et al., ibid. 44, 985 (1961).
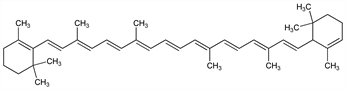
Properties
Deep purple prisms, polyhedra from petr ether or from benzene + methanol, mp 187.50° (evacuated tube). [α]18643 +385° (c = 0.08 in benzene). Absorption max (CHCl3): 485, 454 nm. More sol than β-carotene. Freely sol in carbon disulfide, chloroform; sol in ether, benzene. Slightly sol in petr ether, alcohol. 100 ml hexane dissolves 294 mg at 0°. Practically insol in water, acids, alkalies. Absorbs oxygen from the air, giving rise to inactive colorless oxidation products. The oxidation in light is autocatalytic. Store in darkness in sealed ampoules and at low temp (-20°C).Therapeutic Category
Vitamin A precursor.
Therapeutic Category (Veterinary)
Vitamin A precursor for all species except cats.Keywords
Vitamin/Vitamin Source; Vitamin A