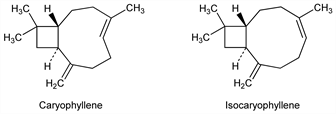
1875. Caryophyllene
Nomenclature
Description and references
Sesquiterpenoid occurring in many essential oils and especially in clove oil, the oils from stems and flowers of Syzygium aromaticum (L.) Merrill & Perry (Jambrosa caryophyllus Niedenzu; Eugenia caryophyllata Thunb.), Myrtaceae. Occurs in nature as a mixture with isocaryophyllene and α-caryophyllene (humulene, q.v.). Isolation of mixture: Schreiner, Kremers, Pharm. Arch. 2, 273, 293 (1899). Existence of isomers: Deussen, Ann. 356, 1 (1907). Structural studies: A. Aebi et al., J. Chem. Soc. 1953, 3124; G. R. Ramage, R. Whitehead, ibid. 1954, 4336. Abs config: D. H. R. Barton, A. Nickon, ibid. 4665. Total synthesis of racemic trans/cis-forms: E. J. Corey et al., J. Am. Chem. Soc. 86, 485 (1964). Rearrangement to isocaryophyllene: Rachlin, DE 2044018 (1971 to I.F.F.), C.A. 75, 49364j (1971). Reviews: Simonsen, The Terpenes vol. III (University Press, Cambridge, 1952) pp 39-71; Barton, de Mayo, Q. Rev. Chem. Soc. 11, 197 (1957); Halsall, ibid. 16, 101 (1962).