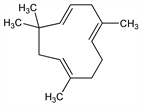
4753. Humulene
Nomenclature
Description and references
Sesquiterpenoid isomer of caryophyllene, q.v. occurring in many essential oils, especially oil of hops (Humulus lupulus L. Moraceae) and leaves of Lindera strychnifolia (F.) Will Lauraceae. Occurs in nature as a mixture with β-humulene. Isolation of mixture: A. C. Chapman, J. Chem. Soc. 67, 54, 780 (1895). Identity with α-caryophyllene: F. Sorm et al., Collect. Czech. Chem. Commun. 14, 693, 699, 716 (1949). Structure: F. Sorm et al., ibid. 19, 570 (1954). Stereochemistry: A. T. McPhail, G. A. Sim, J. Chem. Soc. B 1966, 112. Synthesis: E. J. Corey, E. Hanamaka, J. Am. Chem. Soc. 89, 2758 (1967). Stereoselective synthesis: Y. Kitagawa et al., ibid. 99, 3864 (1977); E. J. Corey et al., Tetrahedron Lett. 34, 3675 (1993). Chromatographic conversion to β-humulene: V. Benesova et al., Collect. Czech. Chem. Commun. 26, 1832 (1961). Reviews: F. Sorm in Fortschr. Chem. Org. Naturst. 19, 1-32 (1961); Rodd's Chemistry of Carbon Compounds vol. IIC, S. Coffey, Ed., (Elsevier, New York, 2nd ed., 1969) pp 282-283.