1920. Cefdinir
Nomenclature
CAS number: 91832-40-5
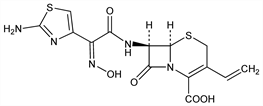
(6R,7R)-7-[[(2Z)-(2-Amino-4-thiazolyl)(hydroxyimino)acetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid; syn-7-[2-(2-amino-4-thiazolyl)-2-hydroxyiminoacetamido]-3-vinyl-3-cephem-4-carboxylic
acid; FK-482; BMY-28488; Omnicef (Abbott). C14H13N5O5S2; mol wt 395.41.
C 42.53%, H 3.31%, N 17.71%, O 20.23%, S 16.22%.
Description and references
Cephalosporin antibiotic structurally similar to cefixime, q.v. Prepn: T. Takaya et al., BE 897864; eidem, US 4559334 (1984, 1985 both to Fujisawa); Y. Inamoto et al., J. Antibiot. 41, 828 (1988); H. Kamachi et al., ibid. 1602. Improved prepn: M. González et al., Farmaco 58, 409 (2003). Clinical trial in children with acute otitis media: S. L. Block et al., Pediatr. Infect. Dis. J. 23, 834 (2004). Review of pharmacokinetics and in vitro antimicrobial activity: D. R. P. Guay, ibid. 19, S141-S146 (2000); of antibacterial activity, pharmacology and clinical efficacy: C. M. Perry, L. J. Scott, Drugs 64, 1433-1464 (2004).
Properties
White to slightly brownish-yellow solid, mp 170° (dec). uv max (pH 7 phosphate buffer): 223, 286 nm (ε 17400, 19700). pKa 9.70. Slightly sol in dilute HCl; sparingly sol in 0.1 M pH 7.0 phosphate buffer.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins