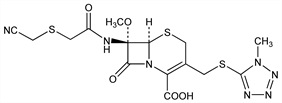
1926. Cefmetazole
Nomenclature
Description and references
Semi-synthetic antibiotic derived from cephamycin C, q.v. Prepn: H. Nakao et al., DE 2455884 (1975 to Sankyo), C.A. 83, 97330g (1975); eidem, J. Antibiot. 29, 554 (1976). See also: J. E. Dolfini, US 3920639 (1975 to Squibb). In vitro study: J. V. Uri et al., ibid. 31, 82 (1978). Immunological study: M. Iwata, T. Matuhasi, Jpn. J. Exp. Med. 48, 401 (1978). Absorption, distribution, excretion, metabolism in various species: H. Shindo et al., Chemotherapy (Tokyo) 27, Suppl. 1, 64 (1979). Pharmacology: S. Kobayashi et al., ibid. 26, Suppl. 5, 115 (1978). Toxicological studies: H. Masuda et al., Sankyo Kenkyusho Nempo 30, 112 (1978), C.A. 90, 180268h (1979). Comparative clinical study: M. Nishida et al., J. Antibiot. 32, 1319 (1979).
Derivative
Sodium salt.Nomenclature
Properties
White solid. Very sol in water, methanol, sol in acetone. Practically insol in chloroform. LD50 i.v. in rats: >5000 mg/kg (Masuda).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephamycins