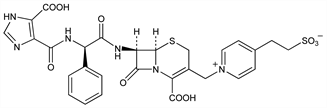
1938. Cefpimizole
Nomenclature
Description and references
Third generation injectable cephalosporin antibiotic. Prepn: N. Yasuda et al., DE 2826546; eidem, US 4217450 (1979, 1980 both to Ajinomoto); N. Yasuda et al., J. Antibiot. 36, 242 (1983). In vitro antibacterial activity and β-lactamase stability: H. C. Neu, P. Labthavikul, Antimicrob. Agents Chemother. 24, 375 (1983). Potentiating effect on phagocyte functions: H. Ohnishi et al., ibid. 23, 874 (1983). Toxicity study: S. Hashimoto et al., Toxicol. Lett. 23, 135 (1984). Therapeutic efficacy in mice: Y. Obana et al., J. Antimicrob. Chemother. 16, 727 (1985). HPLC determn in human plasma and urine: D. B. Lakings, J. M. Wozniak, J. Chromatogr. 308, 261 (1984). Pharmacokinetics in humans: D. B. Lakings et al., Antimicrob. Agents Chemother. 29, 271 (1986). Efficacy and tolerance in gonorrhea in men: E. T. Sandberg et al., ibid. 849.
Derivative
Monosodium salt.Nomenclature
Properties
[α]D20 -28.2° (c = 0.5 in water). uv max (water): 257 nm (ε 22400). Sol in water. LD50 in male, female mice, male, female rats (g/kg): 2.7, 2.9, 4.2, 3.5 i.v.; 8.2, 6.8, 12.2, 11.5 s.c.; all >15.0 orally (Hashimoto).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins