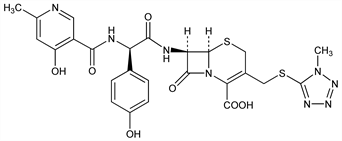
1939. Cefpiramide
Nomenclature
Description and references
Broad spectrum semi-synthetic cephalosporin antibiotic. Prepn: H. Yamada et al., BE 833063; eidem, US 4156724; US 4160087 (1975, 1979, 1979 all to Sumitomo); eidem, JP Kokai 79 119497 (1979 to Sumitomo), C.A. 92, 11042j (1980); I. Isaka et al., JP Kokai 79 30197 (1979 to Yamanouchi), C.A. 91, 57037a (1979); JP Kokai 84 65094 (1984 to Sumitomo), C.A. 101, 110641w (1984). Prepn, NMR data, antibacterial activity: H. Yamada et al., J. Antibiot. 36, 522, 543 (1983). Pharmacokinetics: H. Matsui et al., Antimicrob. Agents Chemother. 22, 213 (1982); K. Sata et al., ibid. 26, 578 (1984). Metabolism: H. Imasaki et al., ibid. 24, 42 (1983). In vitro activity: H. Wexler et al., ibid. 25, 162 (1984); M. A. Pfaller et al., ibid. 368. Comparison with other cephalosporins: N. J. Khan et al., ibid. 26, 585 (1984). Series of articles on in vitro, in vivo activity, determn in body fluids, pharmacokinetics, clinical studies: Chemotherapy (Tokyo) 31, Suppl. 1, 1-842 (1983).
Properties
Yellow crystals, mp 213-215° (dec).Derivative
Sodium salt.Nomenclature
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins