1941. Cefpodoxime Proxetil
Nomenclature
Description and references
Broad spectrum, orally absorbed third generation cephalosporin, ester prodrug of the active free acid metabolite, cefpodoxime. Prepn: H. Nakao et al., EP 49118; eidem, US 4486425 (1982, 1984 both to Sankyo). Prepn, pharmacokinetics and NMR analysis: K. Fujimoto et al., J. Antibiot. 40, 370 (1987). In vitro and in vivo antibacterial activity: Y. Utsui et al., Antimicrob. Agents Chemother. 31, 1085 (1987). In vitro antibacterial spectrum and susceptibility testing of free acid: R. N. Jones, A. L. Barry, ibid. 32, 443 (1988). Isomer-specific HPLC determn in biological fluids: V. K. Kakumanu et al., J. Chromatogr. B 835, 16 (2006). Characterization of isomers: eidem, Eur. J. Pharm. Biopharm. 64, 255 (2006). Series of articles on antibacterial spectrum, pharmacokinetics, toxicology and clinical studies: Chemotherapy (Tokyo) 36, Suppl. 1, 1-1126 (1988). Symposium on pharmacokinetics and clinical efficacy: J. Antimicrob. Chemother. 26, Suppl. E, 1-101 (1990). Review of chemistry, antibacterial activity, toxicity and clinical studies: H. Nakao et al., Sankyo Kenkyusho Nempo 39, 1-44 (1987), C.A. 109, 27503x (1988). Comprehensive review: W. M. Todd, Int. J. Antimicrob. Agents 4, 37-62 (1994).
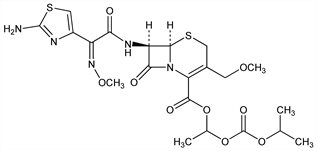
Properties
White to slightly brownish white powder; bitter taste and no odor or slight unique smell. No defined mp. Very sol in acetonitrile, methanol; freely sol in dehydrated ethanol; slightly sol in ether; very slightly sol in water. pKa 3.20±0.13. Partition coefficient (octanol/water): 0.08 (pH 1.2); 1.53 (pH 5); 1.50 (pH 9). Partition coefficient (chloroform/water): 1.60 (pH 1.2); 3.08 (pH 5); 3.18 (pH 9). LD50 in male, female mice, male, female rats (mg/kg): >10000, >10000, >2000, >2000 s.c., 3502, 2535, >4000, >4000 i.p.; >8000, >8000, >4000, >4000 orally (Nakao, 1988).Derivative
Free acid.Nomenclature
Derivative
Free acid sodium salt.Nomenclature
Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins