1942. Cefprozil
Nomenclature
Description and references
Semisynthetic oral cephalosporin consisting of ≈90:10 Z/E isomeric mixture. Prepn and antibacterial activity: H. Hoshi et al., DE 3402642; eidem, US 4520022 (1984, 1985 both to Bristol-Myers); T. Naito et al., J. Antibiot. 40, 991 (1987). Separation of isomers: M. A. Kaplan et al., US 4727070 (1988 to Bristol-Myers). Comparative in vitro antibacterial spectrum and β-lactamase stability: R. N. Jones et al., Diagn. Microbiol. Infect. Dis. 9, 11 (1988). Clinical pharmacokinetics and tissue penetration: R. H. Barbhaiya et al., Antimicrob. Agents Chemother. 34, 1204 (1990). Comparative clinical trials: A. Iravani, ibid. 35, 1940 (1991); A. G. Arguedas et al., Pediatr. Infect. Dis. J. 10, 375 (1991). HPLC determn in plasma and urine: W. C. Shyu et al., Pharm. Res. 8, 992 (1991). Symposium on clinical use: Clin. Infect. Dis. 14, Suppl. 2, S183-S272 (1992).
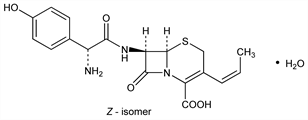
Derivative
Z-Form.Nomenclature
Properties
Isolated as the hemihydrate, crystals from acetone, mp 218-220° (dec). uv max (pH 7 phosphate buffer): 228, 279 nm (ε 12300, 9800).Derivative
E-Form.Nomenclature
Properties
Colorless prisms from methanol, mp 230° (dec). uv max (pH 7 phosphate buffer): 228, 292 nm (ε 13000, 16900).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins