1973. Cephaeline
Nomenclature
CAS number: 483-17-0
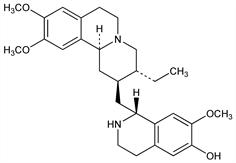
7′,10,11-Trimethoxyemetan-6′-ol; desmethylemetine; dihydropsychotrine. C28H38N2O4; mol wt 466.61.
C 72.07%, H 8.21%, N 6.00%, O 13.72%.
Description and references
Next to emetine the most important alkaloid of ipecac, the ground roots of Uragoga ipecacuanha (Brot.) Baill. [Cephaelis ipecacuanha (Brot.) A. Rich.], Rubiaceae: Carr, Pyman, J. Chem. Soc. 105, 1591 (1914); Hesse, Ann. 405, 1 (1914). Structure: Pailer, Porschinski, Monatsh. Chem. 80, 101 (1949). Stereochemistry: Van Tamelen et al., J. Am. Chem. Soc. 79, 4817 (1957). Partial synthesis: A. K. Garg, J. R. Gear, Tetrahedron Lett. 50, 4377 (1969). Biosynthetic studies: eidem, ibid. 1968, 141. HPLC determn in biological fluids: D. J. Crouch et al., J. Anal. Toxicol. 8, 63 (1984). Review: M. Janot in Manske, Holmes, The Alkaloids vol. 3 (Academic Press, New York, 1953) pp 363-394.
Properties
Needles from ether. Faintly bitter taste. mp 115-116°. [α]D20 -43.4° (c = 2 in chloroform). Practically insol in water. Freely sol in dil hydrochloric acid, dil sulfuric acid, acetic acid, methanol, ethanol, acetone, chloroform. Less sol in ether, petr ether.Derivative
Dihydrochloride heptahydrate.C28H40Cl2N2O4.7H2O; mol wt 665.64.
C 50.52%, H 8.18%, Cl 10.65%, N 4.21%, O 26.44%.
Properties
Prisms, sinters at 245°, slowly melts up to 270°. [α]D20 +25.0° (c = 2). Sol in water. Solns turn yellow. Less sol in alc, acetone, chloroform. Practically insol in benzene, ligroin.Derivative
Dihydrobromide heptahydrate.C28H40Br2N2O4.7H2O; mol wt 754.54.
C 44.57%, H 7.21%, Br 21.18%, N 3.71%, O 23.32%.
Properties
Prisms from dil hydrobromic acid, sinters at 266°, slowly melts up to 293°. Sol in water; moderately sol in alcohol, acetone. Practically insol in benzene.Derivative
Methyl ether see Emetine.Therapeutic Category
Emetic; antiamebic.
Therapeutic Category (Veterinary)
Has been used as an emetic and expectorant.Keywords
Antiamebic; Emetic