1974. Cephalexin
Nomenclature
CAS number: 15686-71-2
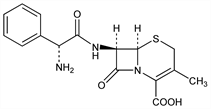
(6R,7R)-7-[[(2R)-Aminophenylacetyl]amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid; 7-(d-α-aminophenylacetamido)desacetoxycephalosporanic
acid; 7-(d-2-amino-2-phenylacetamido)-3-methyl-Δ3-cephem-4-carboxylic acid; Cefadros (Proter); Cefaloto (Lifepharma); Cefanex (Apothecon); Cefaseptin (Chassot); Cex (Nippon Glaxo); Derantel (Nippon Chemiphar); Efalexin (Maipe); Farexin (Lafare); Fergon 500 (Alfar); Garasin (Wakamoto); Ibilex (IBI); Iwalexin (Iwaki); Larixin (Toyama); Lexibiotico (Llano); Llonexina (Castillon); Madlexin (Meiji); Mamalexin (Showa Yakuhin); Mecilex (C.E.P.A.); Ohlexin (Ohta); Oracocin (Tobishi); Rinesal (Kissei); Sencephalin (Takeda); Sintolexyn (ISF-Italseber); Syncl (Toyo Jozo); Taicelexin (Taiyo); Tokiolexin (Isei); Xahl (SS Pharm.). C16H17N3O4S; mol wt 347.39.
C 55.32%, H 4.93%, N 12.10%, O 18.42%, S 9.23%.
Description and references
Semi-synthetic cephalosporin antibiotic. Prepn: R. B. Morin, B. G. Jackson, US 3275626; US 3507861 (1966, 1970 both to Lilly); Ryan et al., J. Med. Chem. 12, 310 (1969). Pharmacology and toxicology: Muggleton et al., Antimicrob. Agents Chemother. 1968, 353; Kind et al., Welles et al., ibid. 361, 489. Comprehensive description: L. P. Marrelli, Anal. Profiles Drug Subs. 4, 21-46 (1975). Clinical pharmacology, efficacy, adverse reactions: Postgrad. Med. J. 59, Suppl. 5, 1-56 (1983).
Properties
Crystals. uv max: 260 nm (ε 7750). pKa 5.2, 7.3.Derivative
Monohydrate.Nomenclature
CAS number: 23325-78-2
Cefa-Iskia (Iskia); Cefibacter (Rubio); Ceporex (GSK); Ceporexin (GSK); Keforal (Lilly); Keflet (Lilly); Keflex (Lilly); Oracef (Lilly); Ortisporina (Turro); Sartosona (Sastre); Servispor (Servipharm). Properties
LD50 in mice, rats (g/kg): 1.6-4.5, >5.0 orally; 0.4-1.3, >3.7 i.p. (Welles).Derivative
Monohydrochloride monohydrate.Nomenclature
CAS number: 105879-42-3
LY-061188; Keftab (Lilly). C16H17N3O4S.HCl.H2O; mol wt 401.87.
C 47.82%, H 5.02%, N 10.46%, O 19.91%, S 7.98%, Cl 8.82%.
Derivative
Sodium salt.Nomenclature
CAS number: 38932-40-0
Alfaspoven (Alfa). C16H16N3NaO4S; mol wt 369.37.
C 52.03%, H 4.37%, N 11.38%, Na 6.22%, O 17.33%, S 8.68%.
Derivative
Pivalate ester see Pivcefalexin.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins