1979. Cephaloridine
Nomenclature
CAS number: 50-59-9
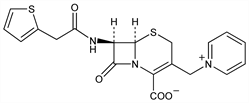
1-[[(6R,7R)-2-Carboxy-8-oxo-7-[(2-thienylacetyl)amino]-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]pyridinium
inner salt; 1-[(7′-β-[2-(2-thienyl)acetamido]-8′-oxo-1′-aza-5′-thiabicyclo[4.2.0]oct-2′-en-3′-yl)methyl]pyridinium-2′-carboxylate; N-[7-[(2-thienyl)acetamido]ceph-3-em-3-ylmethyl]pyridinium-4-carboxylate; N-[7-(2′-thienylacetamidoceph-3-ylmethyl)]pyridinium-2-carboxylate; cefaloridin; Ceflorin (Glaxo); Cepaloridin (Glaxo); Cer (Nippon Glaxo); Faredina (Lafare); Ceporan (Glaxo); Ceporin (Glaxo); Cilifor (C.E.P.A.); Intrasporin (Torlan); Keflodin (Lilly); Keflordin (obsolete) (Lilly); Lloncefal (Castillon); Sefacin (Schering); Cepalorin; Deflorin (Glaxo); Kefspor (Lilly); Loridine (Lilly); Ampligram (Hermes); Floridin (Coli). C19H17N3O4S2; mol wt 415.49.
C 54.92%, H 4.12%, N 10.11%, O 15.40%, S 15.43%.
Description and references
Semi-synthetic cephalosporin antibiotic. Prepn: FR 1384197 (1965 to Glaxo), C.A. 63, 11591c (1965); E. H. Flynn, US 3449338 (1969 to Lilly). Toxicity studies: Atkinson et al., Toxicol. Appl. Pharmacol. 8, 398 (1966).
Properties
Crystals. Soluble in water. Aq solns are slightly acid (pH 4.5-5); [α]D +47.7° (c = 1.25 in water). uv max: 239, 252 nm (ε 15160, 13950). pKa 3.2. Aq solns (20% w/v) are stable for four weeks at 4° in the dark. Exposure to sunlight causes discoloration. LD50 mice, rats (g/kg): >15, 2.5-4 orally; in monkeys (g/kg): >0.2 i.m. (Atkinson).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins