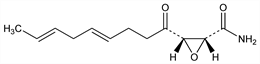
2004. Cerulenin
Nomenclature
Description and references
Antifungal antibiotic isolated from Cephalosporium caerulens; Acryocylindrium oryzae; Helicoceras oryzae: T. Hata et al., Jpn. J. Bacteriol. 15, 1075 (1960); Matsumae et al., J. Antibiot. 16A, 236, 239 (1963); Sano et al., ibid. 20A, 344 (1967); Furuya, Shirasaka, JP 70 21638 (1970 to Sankyo), C.A. 73, 108271k (1970). Biological characteristics: Matsumae et al., J. Antibiot. 17A, 1 (1964). Structure: Omura et al., ibid. 20A, 349 (1967). Abs config: eidem, Chem. Pharm. Bull. 17, 2361 (1969); Arison, Omura, J. Antibiot. 27, 28 (1974). Stereoselective synthesis of (+)- and (-)-tetrahydrocerulenin and corrected abs config of (+)-cerulenin: H. Ohrui, S. Emoto, Tetrahedron Lett. 1978, 2095; J.-R. Pougny, P. Sinay, ibid. 3301. Stereoselective synthesis of the (+)-form from d-glucose: N. Sueda et al., ibid. 1979, 2039; M. Pietraszkiewicz, P. Sinay, ibid. 4741. Interrupts yeast-type fungi growth by inhibiting the biosynthesis of sterols and fatty acids. Mechanism of action studies: Nomura et al., J. Biochem. 71, 783 (1972); eidem, J. Antibiot. 25, 365 (1972); see also D. Vance, Biochem. Biophys. Res. Commun. 48, 649 (1972). Total synthesis of (±)-cerulenin: R. K. Boeckman, Jr., E.W. Thomas, J. Am. Chem. Soc. 99, 2805 (1977); A. A. Jakubowski et al., Tetrahedron Lett. 1977, 2399; E. J. Corey, D. R. Williams, ibid. 3847; K. Mikami et al., Chem. Lett. 1981, 1721; A. A. Jakubowski et al., J. Org. Chem. 47, 1221 (1982).