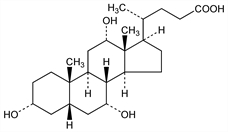
2203. Cholic Acid
Nomenclature
CAS number: 81-25-4
(3α,5β,7α,12α)-3,7,12-Trihydroxycholan-24-oic acid; cholalic acid; 17β-(1-methyl-3-carboxypropyl)etiocholane-3α,7α,12α-triol; Colalin. C24H40O5; mol wt 408.57.
C 70.55%, H 9.87%, O 19.58%.
Description and references
Occurs in conjugation with glycine or taurine in bile of most vertebrates. Extraction from beef bile: Wieland, Siebert, Z. Physiol. Chem. 262, 1 (1939); laboratory procedure: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 360. Structure and synthesis: Fieser, Fieser, Steroids (Reinhold, New York, 1959).
Derivative
Monohydrate.Properties
Plates from dil acetic acid. Bitter taste with sweetish aftertaste. When anhydr, mp 198°. [α]D20 +37° (c = 0.6 in alcohol). pK = 6.4. Not precipitated by digitonin. Soly at 15° in water 0.28 g/l; in alcohol 30.56 g/l; in ether 1.22 g/l; in chloroform 5.08 g/l; in benzene 0.36 g/l; in acetone 28.24 g/l; in glacial acetic acid 152.12 g/l. Sol in solns of alkali hydroxides or carbonates.Derivative
Methyl ester.C25H42O5; mol wt 422.60.
C 71.05%, H 10.02%, O 18.93%.
Properties
Crystals from 95% alcohol + water, mp 155-156°.Derivative
Ethyl ester.C26H44O5; mol wt 436.62.
C 71.52%, H 10.16%, O 18.32%.
Properties
Crystals from ethyl acetate + petr ether, mp 162-163°.Derivative
Sodium salt.Nomenclature
Sodium cholate.C24H39NaO5; mol wt 430.55.
C 66.95%, H 9.13%, Na 5.34%, O 18.58%.
Properties
Crystals. Soly in water at 15°: >568.9 g/l.Caution
The names “sodium cholate” and “sodium choleate” are sometimes used for mixtures of bile salts. The term “sodium choleate” is to be preferred for bile salts, see Ox Bile Extract.Note
The property of forming molecular compds is common to bile acids. For instance a blue molecular compd (C24H40O5I)4.KI.H2O, may be prepd by mixing an alcoholic soln of cholic acid and a soln of iodine in aq potassium iodide: Barger, Field, J. Chem. Soc. 101, 1404 (1912).Therapeutic Category
Choleretic.
Keywords
Choleretic