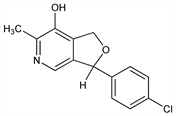
2264. Cicletanine
Nomenclature
Description and references
Furopyridine derivative with antihypertensive and diuretic properties. Prepn: A. Esanu, BE 891797; idem, US 4383998 (1982, 1983 both to Soc. Conseils Recher. Sci.). Studies on mechanism of action: P. Braquet et al., Lancet 1, 1218 (1983); G. R. Elliott et al., Thromb. Res. 33, 549 (1984). Cardiovascular pharmacology in dogs: R. Jouve et al., J. Cardiovasc. Pharmacol. 8, 208 (1986). In vitro comparison of antihistaminic activity of isomers: P. Schoeffter et al., Eur. J. Pharmacol. 136, 235 (1987). HPLC determn in biological fluids: G. Cuisinaud et al., J. Chromatogr. 341, 97 (1985). Effect on human prostaglandin metabolism: P. Guinot, J. C. Fr"olich, Arzneim.-Forsch. 35, 1714 (1985). Effect on potassium ion transport in hypertensive patients: R. Garay et al., J. Hypertens. 4, Suppl. 5, S208 (1986). Pharmacokinetics in humans: J. M. Lize et al., Therapie 42, 399 (1987).
Derivative
Hydrochloride.Nomenclature
Properties
White crystals, mp 219-228°. Insol in water.Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive