2356. Clindamycin
Nomenclature
CAS number: 18323-44-9
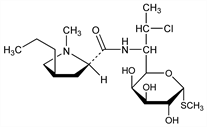
Methyl 7-chloro-6,7,8-trideoxy-6-[[[(2S,4R)-1-methyl-4-propyl-2-pyrrolidinyl]carbonyl]amino]-1-thio-l-threo-α-d-galacto-octopyranoside; 7(S)-chloro-7-deoxylincomycin; 7-deoxy-7(S)-chlorolincomycin; clinimycin (rescinded); U-21251. C18H33ClN2O5S; mol wt 424.98.
C 50.87%, H 7.83%, Cl 8.34%, N 6.59%, O 18.82%, S 7.55%.
Description and references
Semi-synthetic antibiotic prepd from lincomycin, q.v. Prepn: Magerlein et al., Antimicrob. Agents Chemother. 1966, 727. Manuf process: R. D. Birkenmeyer, US 3475407 (1969 to Upjohn). Synthesis and structure: R. D. Birkenmeyer, F. Kagan, J. Med. Chem. 13, 616 (1970). Stability studies: Oesterling, J. Pharm. Sci. 59, 63 (1970). Activity studies: McGehee et al., Am. J. Med. Sci. 256, 279 (1968); D. A. Leigh, J. Antimicrob. Chemother. 7(suppl. A), 3 (1981). Toxicology: J. E. Gray et al., Toxicol. Appl. Pharmacol. 21, 516 (1972). Comprehensive description of the hydrochloride: L. W. Brown, W. F. Beyer, Anal. Profiles Drug Subs. 10, 75-91 (1981).
Properties
Yellow, amorphous solid. [α]D +214° (chloroform).Derivative
Hydrochloride.Nomenclature
CAS number: 21462-39-5;
58207-19-5
(monohydrate)
Antirobe (Pharmacia & Upjohn); Clinsol (Virbac); Clintabs (Virbac); Dalacin C (Pharmacia & Upjohn); Sobelin (Pharmacia). C18H33ClN2O5S.HCl; mol wt 461.44.
C 46.85%, H 7.43%, Cl 15.37%, N 6.07%, O 17.34%, S 6.95%.
Properties
Various hydrated forms exist. White crystals from ethanol-ethyl acetate, mp 141-143° (anhydrous). [α]D +144° (H2O). pKa 7.6. Sol in water, pyridine, ethanol, DMF. LD50 in mice (mg/kg): 245 i.v.; 361 i.p.; 2618 orally (Gray).Derivative
2-Dihydrogen phosphate.Nomenclature
CAS number: 24729-96-2
U-28508; Cleocin (Pharmacia & Upjohn); Dalacin T (Pharmacia & Upjohn); Klimicin (Lek); Zindaclin (Epitan). C18H34ClN2O8PS; mol wt 504.96.
C 42.81%, H 6.79%, Cl 7.02%, N 5.55%, O 25.35%, P 6.13%, S 6.35%.
Properties
Sol in water.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Lincosamides