2363. Clobetasol
Nomenclature
CAS number: 25122-41-2
(11β,16β)-21-Chloro-9-fluoro-11,17-dihydroxy-16-methylpregna-1,4-diene-3,20-dione. C22H28ClFO4; mol wt 410.91.
C 64.30%, H 6.87%, Cl 8.63%, F 4.62%, O 15.57%.
Description and references
Topical corticosteroid. Prepn: Elks et al., DE 1902340; eidem, US 3721687 (1969, 1973 both to Glaxo). Clinical evaluation of shampoo formulation in severe scalp psoriasis: C. E. M. Griffiths et al., J. Dermatolog. Treat. 17, 90 (2006). Clinical trial of foam formulation in delayed pressure uticaria: G. A. Vena et al., Br. J. Dermatol. 154, 353 (2006). Review of pharmacology and clinical efficacy in skin disorders: E. A. Olsen, R. C. Cornell, J. Am. Acad. Dermatol. 15, 246-255 (1986); of clinical experience of foam formulation in psoriasis: D. C. Reid, A. B. Kimball, Expert Opin. Pharmacother. 6, 1735-1740 (2005).
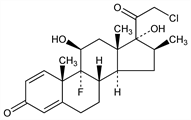
Derivative
17-Propionate.Nomenclature
CAS number: 25122-46-7
GR-2/925; Clobesol (GSK); Clobex (Galderma); Dermovate (GSK); Olux (Connetics); Psorex (GSK); Temovate (GSK). C25H32ClFO5; mol wt 466.97.
C 64.30%, H 6.91%, Cl 7.59%, F 4.07%, O 17.13%.
Properties
White or almost white colorless, crystalline powder, mp 195.5-197°. [α]D +103.8° (c = 1.04 in dioxane). uv max (ethanol): 237 nm (ε 15000). Insol in water.Therapeutic Category
Glucocorticoid; anti-inflammatory; antipsoriatic.
Keywords
Glucocorticoid; Antipsoriatic