2399. Cloprostenol
Nomenclature
CAS number: 40665-92-7
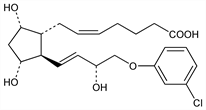
(5Z)-rel-7-[(1R,2R,3R,5S)-2-[(1E,3R)-4-(3-Chlorophenoxy)-3-hydroxy-1-butenyl]-3,5-dihydroxycyclopentyl]-5-heptenoic
acid. C22H29ClO6; mol wt 424.92.
C 62.18%, H 6.88%, Cl 8.34%, O 22.59%.
Description and references
Aryloxymethyl analog of prostaglandin F2α, q.v. Prepn: J. Bowler, DE 2223365 (1972 to ICI), C.A. 78, 110692v (1973); D. Binder et al., Prostaglandins 6, 87 (1974). Synthesis and biological activity: eidem, ibid. 15, 773 (1978). Short synthesis: N. R. A. Beeley et al., Tetrahedron 37, Suppl. 9, 411 (1981). Disposition in the rat and marmoset: G. R. Bourne et al., Xenobiotica 9, 623 (1979). Effect on fertility in cows: N. Baishya et al., Br. Vet. J. 136, 227 (1980); on superovulation, fertilization, egg transport in ewes: D. Whyman, R. W. Moore, J. Reprod. Fertil. 60, 267 (1980).
Derivative
Sodium salt.Nomenclature
CAS number: 55028-72-3
ICI-80996; Estrumate (Mallinckrodt); Planate (Mallinckrodt). C22H28ClNaO6; mol wt 446.90.
C 59.13%, H 6.32%, Cl 7.93%, Na 5.14%, O 21.48%.