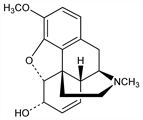
2463. Codeine
Nomenclature
Description and references
Present in opium from 0.7 to 2.5%, depending on the source, but mostly prepd by methylation of morphine, q.v. Discussion of structure and bibliography: Small, Lutz, “Chemistry of the Opium Alkaloids,” in U.S. Public Health Reports Suppl. No. 103 (Washington, 1932). Prepn of (+)-codeine and racemate: Goto, Yamamoto, Proc. Jpn. Acad. 30, 769 (1954), C.A. 50, 1052h (1956); of (-)-form: E. J. Bijsterveld, H. J. Sinnige, Rec. Trav. Chim. 95, 24 (1976); H. C. Beyerman et al., ibid. 97, 127 (1978). Manuf from morphine: W. R. Heumann, Bull. Narc. X, 15 (1958). Facile synthesis from thebaine, q.v.: W. G. Dauben et al., J. Org. Chem. 44, 1567 (1979). Toxicity of the hydrochloride: Eddy, Sumwalt, J. Pharmacol. Exp. Ther. 67, 127 (1939). Comprehensive description of codeine and codeine phosphate, q.v.: F. J. Muhtadi, M. M. A. Hassan, Anal. Profiles Drug Subs. 10, 93-138 (1981).
Derivative
Monohydrate.Properties
Orthorhombic sphenoidal rods or tablets (octahedra) from water or dil alcohol, mp 154-156° (after drying at 80°). Sublimes (when anhydr) at 140-145° under 1.5 mm pressure. Melts to oily drops when heated in an amount of water insufficient for complete soln, crystallizes on cooling. d420 1.32. [α]D15 -136° (c = 2 in alcohol), [α]D15 -112° (c = 2 in chloroform). pK (15°) 6.05; pH of satd aq soln 9.8. One gram dissolves in 120 ml water, 60 ml water at 80°, 2 ml alcohol, 1.2 ml hot alcohol, 13 ml benzene, 18 ml ether, 0.5 ml chloroform; freely sol in amyl alcohol, methanol, dil acids. Almost insol in petr ether or in solns of alkali hydroxides.Derivative
Hydrochloride.Nomenclature
Properties
Dihydrate, small needles, mp ≈280° with some decompn. [α]D22 -108°. One gram dissolves in 20 ml water, 1 ml boiling water, 180 ml alcohol. pH about 5. LD50 s.c. in mice: 300 mg/kg (Eddy, Sumwalt).Derivative
Sulfate.Nomenclature
Properties
Trihydrate, crystals or cryst powder. One gram dissolves in 30 ml water, 6.5 ml water at 80°, 1300 ml alc. Insol in chloroform or ether. pH: 5.0. Store in airtight containers; protect from light.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic; antitussive.
Therapeutic Category (Veterinary)
Analgesic; antitussive.Keywords
Analgesic; Opioids; Phenanthrenes; Antitussive